Synthesis and Biological Activity of Some 3-(4-(Substituted)-piperazin-1-yl)cinnolines
Abstract
:1. Introduction
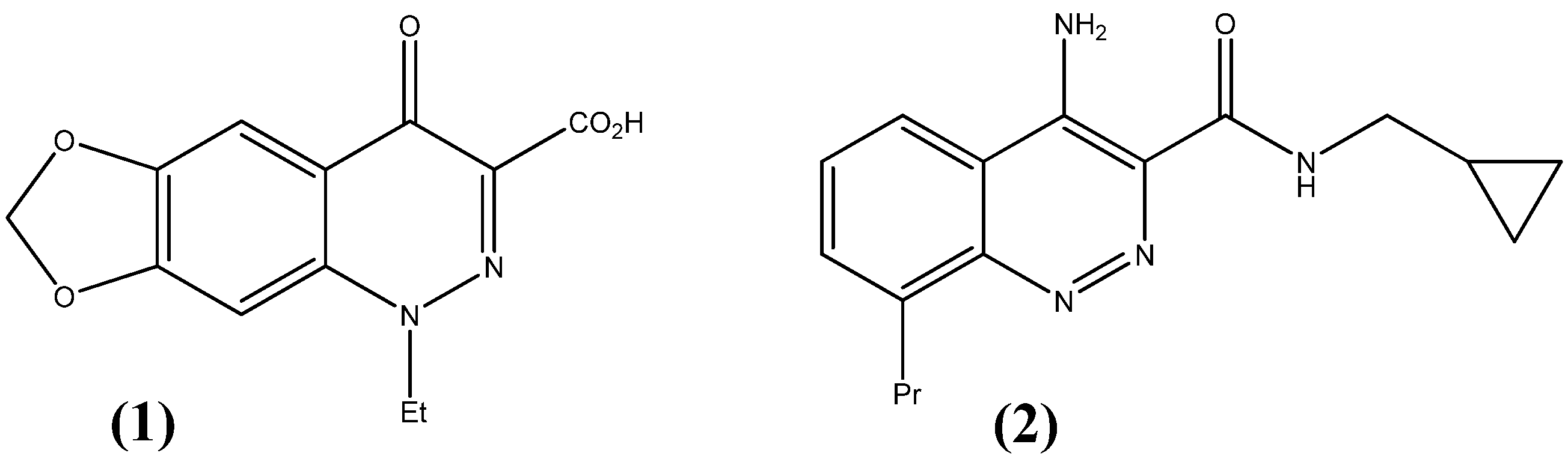
2. Results and Discussion
2.1. Chemistry
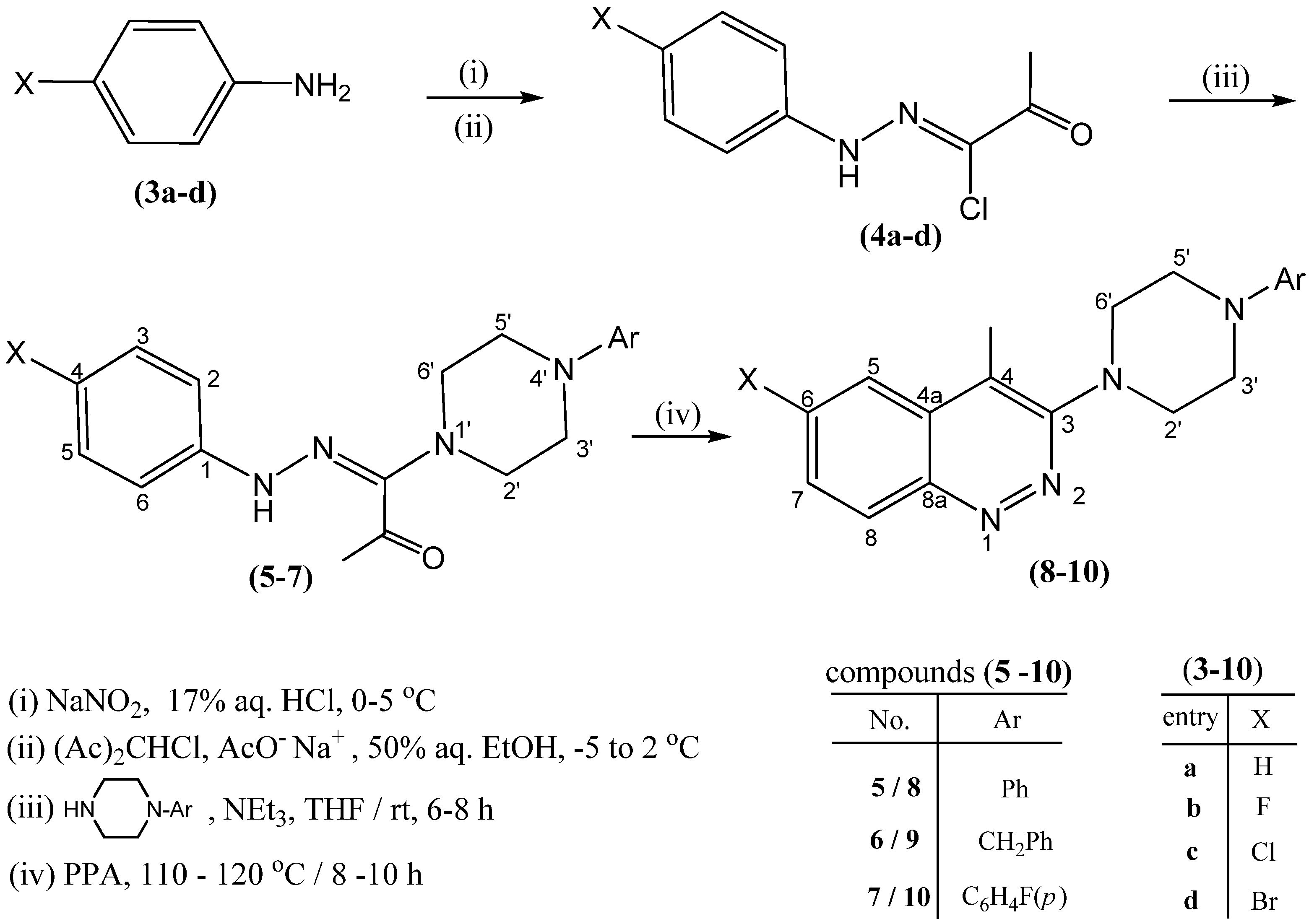
2.2. Antibacterial and Antifungal Activity
2.2.1. Compound Susceptibility Testing by Kirby Bauer Method
| Compound (25 µg/mL) | 8a | 8b | 8c | 8d | 9a | 9b | Nystatin |
| C. albicans ATCC 10231 | NA | NA | NA | NA | 7 ± 0.2 | 7 ± 0.2 | 17 ± 1.5 |
| C. glabirata ATCC 15126 | 8 ± 0.5 | 7 ± 0.1 | 7 ± 0.8 | 7 ± 0.5 | NA | NA | 20 ± 0.5 |
| C. albicans Clinical isolate | 7 ± 0.5 | 7 ± 0.1 | 7 ± 0.5 | 7 ± 0.1 | 8 ± 0.6 | 7 ± 0.2 | 15 ± 1 |
| Compound (25 µg/mL) | 9c | 9d | 10a | 10b | 10c | 10d | Nystain |
| C. albicans ATCC 10231 | NA | 7 ± 0.2 | 8 ± 0.4 | NA | NA | 7 ± 0.5 | 17 ± 1.5 |
| C. glabirata ATCC 15126 | NA | NA | 8 ± 0.5 | 8 ± 0.7 | 8 ± 0.4 | 9 ± 0.3 | 20 ± 0.5 |
| C. albicans Clinical isolate | 7 ± 0.6 | 7 ± 0.5 | 8 ± 0.3 | 7 ± 0.1 | 7 ± 0.1 | 8 ± 0.2 | 15 ± 1 |
2.2.2. Compound Susceptibility Testing by Microbroth Dilution Method
| Entry | 8a | 8b | 8c | 8d | Nystatin | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | |
| C. albicans clinical isolate | 0.4 | 0.9 | 0.3 | 0.6 | 0.4 | 0.9 | 3.0 | 5.0 | 0.008 | 0.03 |
| C.galibrata ATCC 15126 | 0.4 | 0.4 | 0.2 | 0.2 | 0.4 | 0.4 | 3.0 | 3.0 | 0.003 | 0.02 |
| Entry | 9a | 9b | 9c | 9d | Nystatin | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | |
| C .albicans ATCC 10231 | 0.6 | 1.0 | 0.6 | 1.0 | NA | NA | 0.8 | 2.0 | 0.006 | 0.03 |
| C. albicans clinical isolate | 0.2 | 0.3 | 0.6 | 1.0 | 0.08 | 0.2 | 0.4 | 0.8 | 0.008 | 0.03 |
| C. galibrata ATCC 15126 | 0.3 | 0.3 | 0.6 | 1.0 | 0.16 | 0.3 | 0.4 | 0.4 | 0.003 | 0.02 |
2.3. Antitumor Activity
| Compound | 8a | 8b | 8c | 8d | 9a | 9b | 9c | 9d | 10a | 10b | 10c | 10d |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MCF-7% survival | 95 | 43 | 92 | 101 | 99 | 102 | 102 | 94 | 101 | 42 | 74 | 45 |
| Standard deviation | ±2.60 | ±3.53 | ±1.85 | ±1.10 | ±7.02 | ±3.21 | ±2.49 | ±0.71 | ±2.25 | ±1.48 | ±1.84 | ±2.31 |
| Compound | 8b | 10b | 10d | Doxorubicin |
|---|---|---|---|---|
| IC50 MCF-7 (μM) | 5.56 ± 0.30 | 11.79 ± 2.05 | 8.57 ± 0.85 | 0.31 ± 0.01 |
3. Experimental
3.1. General
3.2. General Procedure for the Synthesis of Substituted Piperazin-1-yl amidrazones 5–7
3.3. General Procedure for the Synthesis of 4-methyl-3-[(4-substituted)piperazin-1-yl]cinnolines 8–10
3.4. Biological Activity Test Procedures
3.4.1. Candida Cultures
3.4.2. Compound Susceptibility Testing Disk Diffusion Method/(Kirby Bauer method)
3.4.3. Serial Dilution Method (Broth Microdilution Assay)
3.4.4. Statistical Analyses
3.5. Cell Lines and Cell Culture
3.5.1. Materials and Methods
3.5.2. Cell Proliferation Assay
References and Notes
- Barraja, P.; Diana, P.; Lauria, A.; Passannanti, A.; Almerico, A.M.; Minnei, C.; Longu, S.; Congiu, D.; Musiu, C.; LaColla, P. Indolo[3,2-c]cinnolines with antiproliferative, antifungal, and antibacterial activity. Bioorg. Med. Chem. 1999, 7, 1591–1596. [Google Scholar] [CrossRef]
- Hennequin, L.F.; Thomas, A.P.; Johnstone, C.; Stokes, E.S.; Ple, P.A.; Lohman, J.-J.; Ogilve, D.J.; Dukes, M.; Wedge, S.R.; Curwen, J.O.; et al. Design and structure-activity relationship of a new class of potent VEGF receptor tyrosine kinase inhibitors. J. Med. Chem. 1999, 42, 5369–5389. [Google Scholar]
- Yu, Y.; Singh, S.K.; Liu, A.; Li, T.-K.; Liu, L.F.; La Voie, E.J. Substituted dibenzo[c,h] cinnolines: Topoisomerase I-targeting anticancer agents. Bioorg. Med. Chem. 2003, 11, 1475–1491. [Google Scholar] [CrossRef]
- Nargund, L.; Badiger, V.; Yarnal, S. Synthesis and antimicrobial and anti-inflammatory activities of substituted 2-mercapt-3-(N-aryl)pyrlmido[5,4-c]cinnolin-4-(3H)ones. J. Pharm. Sci. 1992, 81, 365–366. [Google Scholar] [CrossRef]
- Bantick, J.; Hirst, S.; Perry, M.; Phillips, E. Preparation of Fused Pyridazines as Allergy Inhibitors and Antiinflammatories. PCT Int. Appl. WO 9809969, 1998. Chem. Abstr. 1998, 128, 217375. [Google Scholar]
- Rehse, K.; Gonska, H. New pyrimido[5,4-c]cinnolines with antiplatelet activities. Arch. Pharm. 2005, 338, 590–597. [Google Scholar] [CrossRef]
- Ramalingam, P.; Ganapaty, S.; Rao, C.B.; Ravi, T.K. Synthesis of some new coumarino[4,3-b]pyrido[6,5-c]cinnolines as potent antitubercular agents. Ind. J. Heterocycl. Chem. 2006, 15, 359–362. [Google Scholar]
- Gomtsyan, A.; Bayburt, E.K.; Schmidt, R.G.; Zheng, G.Z.; Perner, R.J.; Didomenico, S.; Koenig, J.R.; Turner, S.; Jinkerson, T.; Drizin, I.; et al. Novel transient receptor potential vanilloid 1 receptor antagonists for the treatment of pain: Structure-activity relationships for ureas with quinoline, isoquinoline, quinazoline, phthalazine, quinoxaline, and cinnoline moieties. J. Med. Chem. 2005, 48, 744–752. [Google Scholar]
- Alvarado, M.; Barcelo, M.; Carro, L.; Masaguer, C.F.; Ravina, E. avina, E.Synthesis and biological evaluation of new quinazoline and cinnoline derivatives as potential atypical antipsychotics. Chem. Biodivers. 2006, 3, 106–117. [Google Scholar]
- Abdelrazek, F.M.; Metz, P.; Metwally, N.H.; El-Mahrouky, S.F. Synthesis and molluscicidal activity of new cinnoline and pyrano[2,3-c]pyrazole derivatives. Arch. Pharm. 2006, 339, 456–460. [Google Scholar]
- Heinisch, H.; Frank, H. Pharmacolgically active pyridazine derivatives, Part 1. In Progress in Medicinal Chemistry; Ellis, G.P., West, G.G., Eds.; Elsevier: Amsterdam, The Netherlands, 1990; pp. 1–48. [Google Scholar]
- Coates, W.G. Pyridazines and their condensed derivatives. In Comperhensive Heterocyclic Chemistry; Katritzky, A.R., Rees, C.W., Scriven, F.V., Eds.; Pergamon: London, UK, 1996; pp. 1–91. [Google Scholar]
- Melikian, A.; Schlewer, G.; Chambon, J.P.; Wermuth, C.G. Condensation of muscimol or thiomuscimol with aminopyridazines yields GABA-A antagonists. J. Med. Chem. 1992, 35, 4092–4097. [Google Scholar]
- Haider, N.; Holzer, W. Product class 9: Cinnolines. Sci. Synth. 2004, 16, 251–313. [Google Scholar]
- Abdel-Jalil, R.; El Momani, E.; Hamad, M.; Voelter, W.; Mubarak, M.S.; Smith, B.; Peters, D. Synthesis, antitumor activity, and electrochemical behavior of some piperazinyl amidrazones. Monatsh. Chem. 2010, 141, 251–258. [Google Scholar]
- Al-Rifai, A.A.; Ayoub, M.T.; Shakya, A.K.; Abu Safieh, K.A.; Mubarak, M.S. Synthesis, characterization, and antimicrobial activity of some new coumarin derivatives. Med. Chem. Res. 2011. [Google Scholar] [CrossRef]
- Mustafa, M.S.; El-Abadelah, M.M.; Zihlif, M.A.; Naffa, R.; Mubarak, M.S. Synthesis, and antitumor activity of some N1-(coumarin-7-yl) amidrazones and related congeners. Molecules 2011, 16, 4305–4317. [Google Scholar]
- Phillips, R.R. The Japp-Klingemann reaction. Org. React. 1959, 10, 143–178. [Google Scholar]
- Yao, H.-C.; Resnick, P. Azo-hydrazone conversion. I. The Japp-Klingemann reaction. J. Am. Chem. Soc. 1962, 84, 3514–3517. [Google Scholar]
- Barrett, G.C.; El-Abadelah, M.M.; Hargreaves, M.K. Cleavage of 2-acetyl-2-phenylazopropionanilide and related compounds by boron trifluoride. New Japp-Klingemann reactions. J. Chem. Soc. (C) 1970, 1986–1989. [Google Scholar]
- Hussein, A.Q.; El-Abadelah, M.M.; Al-Adhami, K.; Abushamleh, A. Heterocycles from nitrile imines. Part II. Synthesis and ring-chain tautomerism of 1,2,3,4-tetrahydro-s-tetrazines. Heterocycles 1989, 29, 1163–1170. [Google Scholar] [CrossRef]
- El-Abadelah, M.M; Hussein, A.Q.; Thaher, B.A. Heterocycles from nitrile imines. Part IV. Chiral 4,5-dihydro-1,2,4-triazin-6-ones. Heterocycles 1991, 32, 1879–1895. [Google Scholar] [CrossRef]
- Bauer, A.W.; Kirby, W.M.; Sherries, M.; Truck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar]
- Clinical Laboratory Standards Institute, Performance Standards for Antimicrobial Disk Susceptibility Tests, Approved Standard, 9th ed; CLSI: Wayne, PA, USA, 2006; Volume 26, pp. M2–A9, Number 1.
- National Committee for Clinical Laboratory Standards, Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeasts, Approved standard; NCCLS: Wayne, PA, USA, 1997; pp. M27–A.
- National Committee for Clinical Laboratory Standards, Performance Standards for Antimicrobial Susceptibility Testing: Eleventh Informational Supplement; NCCLS: Wayne, PA, USA, 2003; pp. M100–S11.
- GraphPad Software Home Page. Available online: http://www.graphpad.com (accessed on 15 May 2011).
- Sample Availability: Samples of the compounds 8–10 are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Awad, E.D.; El-Abadelah, M.M.; Matar, S.; Zihlif, M.A.; Naffa, R.G.; Al-Momani, E.Q.; Mubarak, M.S. Synthesis and Biological Activity of Some 3-(4-(Substituted)-piperazin-1-yl)cinnolines. Molecules 2012, 17, 227-239. https://doi.org/10.3390/molecules17010227
Awad ED, El-Abadelah MM, Matar S, Zihlif MA, Naffa RG, Al-Momani EQ, Mubarak MS. Synthesis and Biological Activity of Some 3-(4-(Substituted)-piperazin-1-yl)cinnolines. Molecules. 2012; 17(1):227-239. https://doi.org/10.3390/molecules17010227
Chicago/Turabian StyleAwad, Eman D., Mustafa M. El-Abadelah, Suzan Matar, Malek A. Zihlif, Randa G. Naffa, Ehab Q. Al-Momani, and Mohammad S. Mubarak. 2012. "Synthesis and Biological Activity of Some 3-(4-(Substituted)-piperazin-1-yl)cinnolines" Molecules 17, no. 1: 227-239. https://doi.org/10.3390/molecules17010227




