Synthesis and Antifungal Activities of Some Novel Pyrimidine Derivatives
Abstract
:1. Introduction
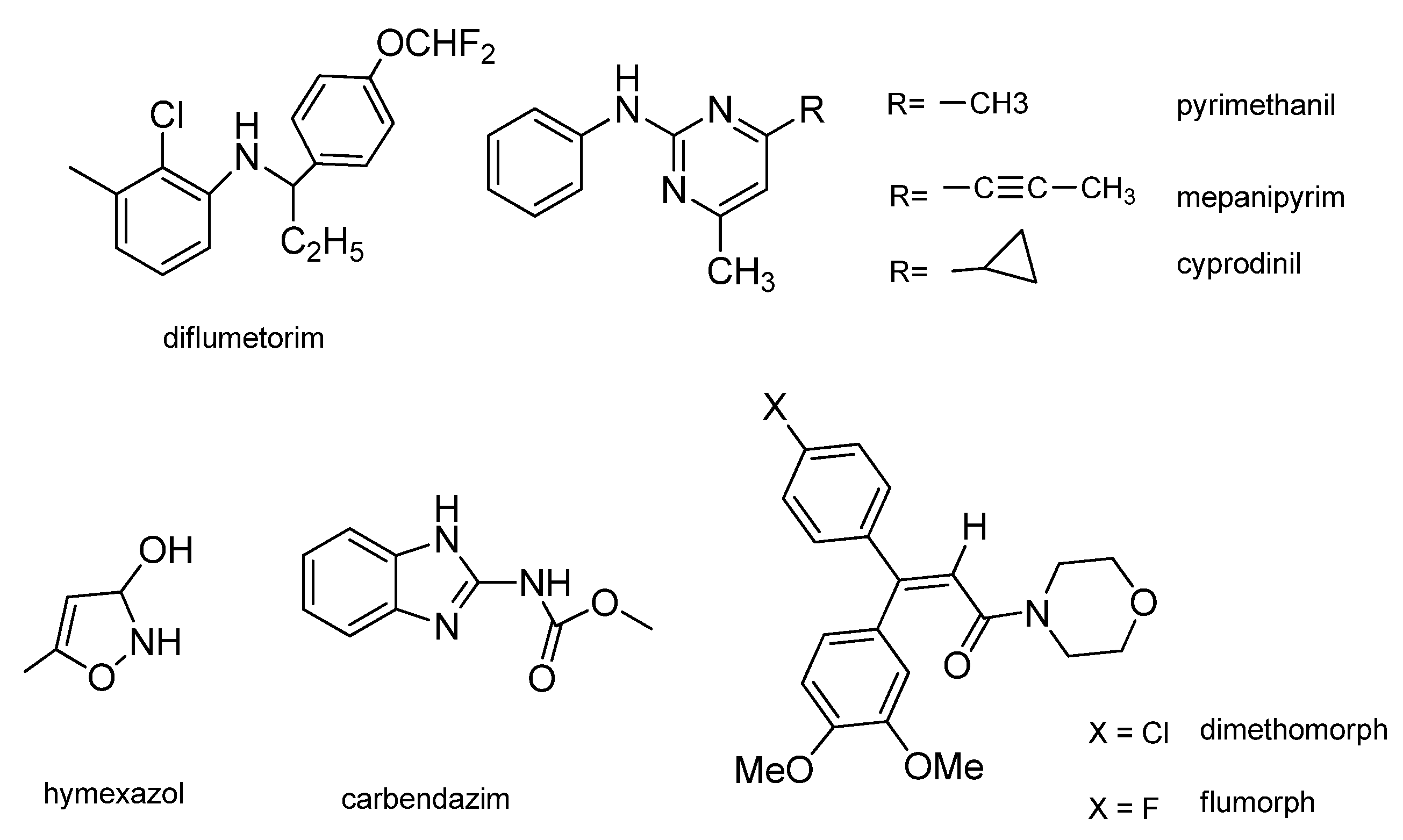

2. Results and Discussion
2.1. Synthesis
2.2. Antifungal Bioassay: Inhibitory Effects on Phytopathogenic Fungi
| Compd. No. | Fungicidal activities (50 μg/mL, inhibition rate %) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AK | AM | AS | BC | BM | CA | GF | GZ | MK | PI | RC | RS | SS | TC | |
| 1a | 45.5 | 21.4 | 41.2 | 29.6 | 34.8 | 13.3 | 20 | 25.9 | 31.3 | 22.7 | 35.0 | 63.5 | 0 | 80.4 |
| 1b | 18.2 | 0 | 0 | 7.4 | 17.4 | 0 | 20 | 14.8 | 31.3 | 0 | 30 | 61.9 | 0 | 3.9 |
| 2a | 18.2 | 0 | 0 | 40.7 | 17.4 | 0 | 20 | 0 | 46.9 | 9.1 | 5.0 | 54.0 | 20 | 2.0 |
| 2b | 31.8 | 21.4 | 5.9 | 37.0 | 17.4 | 13.3 | 26.7 | 29.6 | 46.9 | 0 | 5.0 | 60.3 | 0 | 2.0 |
| 3a | 27.3 | 21.4 | 11.8 | 7.4 | 26.1 | 33.3 | 0 | 7.4 | 21.9 | 0 | 5.0 | 57.1 | 0 | 39.2 |
| 3b | 63.6 | 50 | 35.3 | 25.9 | 39.1 | 46.7 | 40 | 44.4 | 25.0 | 36.4 | 25.0 | 66.7 | 0 | 76.5 |
| 3c | 27.3 | 21.4 | 0 | 3.7 | 13.0 | 46.7 | 13.3 | 22.2 | 0 | 36.4 | 0 | 55.6 | 33.3 | 52.9 |
| 4a | 40.9 | 0 | 0 | 18.5 | 17.4 | 6.7 | 6.7 | 7.4 | 31.3 | 0 | 20 | 61.9 | 0 | 19.6 |
| 4b | 27.3 | 21.4 | 0 | 14.8 | 13.0 | 0 | 46.7 | 14.8 | 46.9 | 18.2 | 0 | 55.6 | 40 | 15.7 |
| 4c | 22.7 | 21.4 | 0 | 33.3 | 26.1 | 6.7 | 53.3 | 40.7 | 37.5 | 13.6 | 0 | 61.9 | 60 | 0 |
| 4d | 31.8 | 28.6 | 0 | 14.8 | 26.1 | 6.7 | 20 | 7.4 | 40.6 | 13.6 | 15.0 | 63.5 | 66.7 | 9.8 |
| Pyri. | 100 | 21.4 | 100 | 96.3 | 91.3 | 100 | 20 | 44.4 | 100 | 22.7 | 40 | 77.8 | 86.7 | 98.0 |
| Flu. | 18.2 | 21.4 | 0 | 7.4 | 17.4 | 0 | 13.3 | 7.4 | 21.9 | 4.5 | 20 | 61.9 | 0 | 9.8 |
| Dim. | 22.7 | 7.1 | 5.9 | 18.5 | 17.4 | 0 | 20 | 0 | 6.3 | 9.1 | 40 | 68.3 | 20 | 23.5 |
| Carb. | 81.8 | 78.6 | 76.5 | 63.0 | 69.6 | 53.3 | 60 | 59.3 | 43.8 | 27.3 | 100 | 100 | 73.3 | 98.0 |
| Hym. | 81.8 | 85.7 | 88.2 | 70.4 | 34.8 | 93.3 | 40 | 33.3 | 50 | 36.4 | 25.0 | 63.5 | 66.7 | 78.4 |
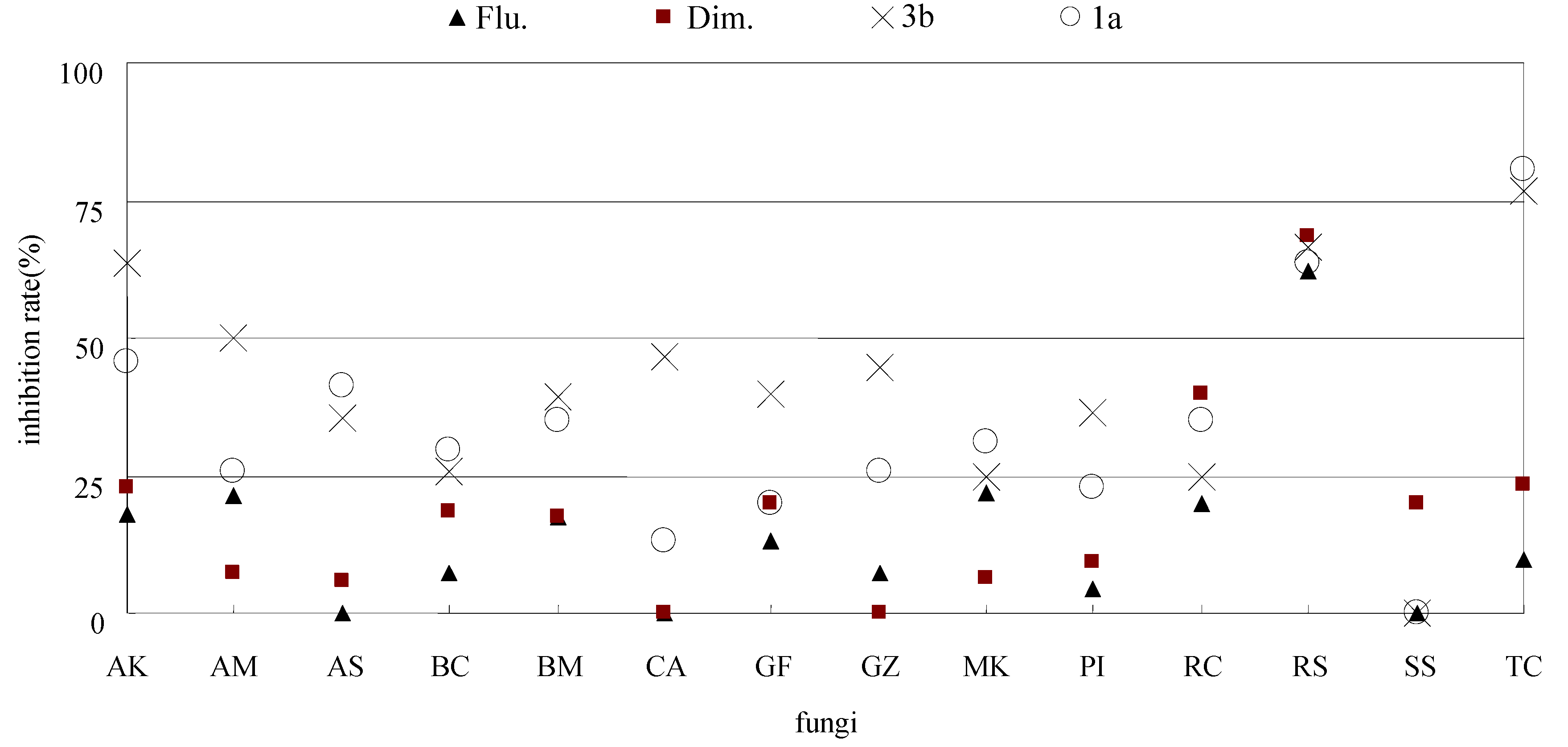
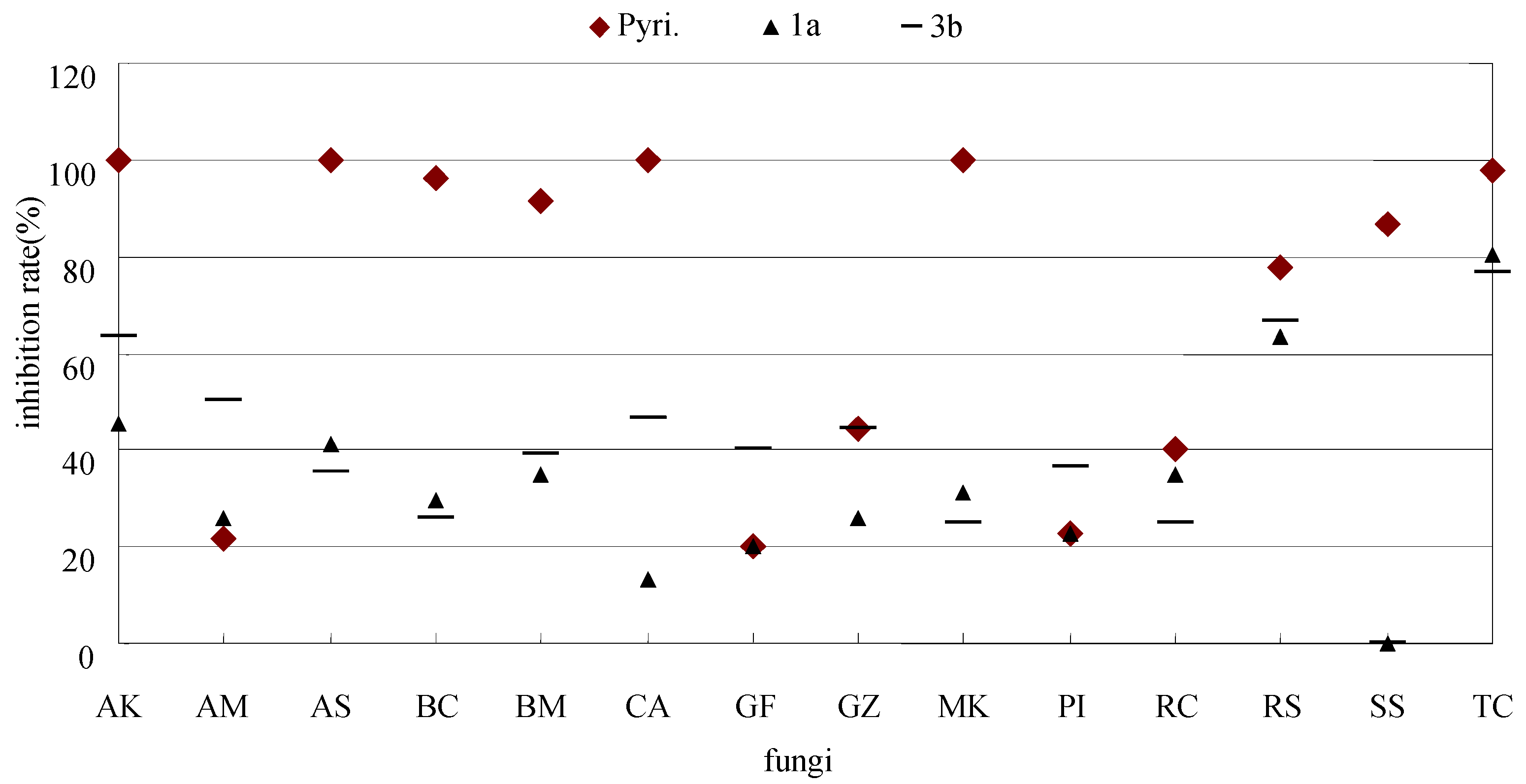
3. Experimental
3.1. General
3.2. Procedures for the Preparation of Title Compounds 1-4
4. Conclusions
Supplementary Materials
Supplementary File 1Acknowledgements
References
- Diánez, F.; Santos, M.; Blanco, R.; Tello, J.C. Fungicide resistance in Botrytis cinerea isolates from strawberry crops in Huelva (southwestern spain). Phytoparasitica 2002, 30, 529–534. [Google Scholar] [CrossRef]
- Stanley, W.M.; Wyokow, R.W.G. Vitamin B1, a growth factor for higher plants. Science 1937, 85, 183–184. [Google Scholar]
- Knight, B.C.J.G. The nutrition of Staphylococcus aureus; nicotinic acid and vitamin B1. Biochem. J. 1937, 31, 731–737. [Google Scholar]
- Guo, J.; Yu, L.; Turro, N.J.; Ju, J. An integrated system for DNA sequencing by synthesis using novel nucleotide analogues. Accounts Chem. Res. 2009, 43, 551–563. [Google Scholar]
- Rashad, A.E.; Shamroukh, A.H.; Abdel-megeid, R.E.; Sayed, H.H.; Abdel-wahed, N.M. Studies on the reactivity of (9-methyl-5,6-dihydronaphtho[1',2':4,5]-thieno[2,3-d]pyrimidin-11-yl)hydrazine towards some reagents for biological evaluation. Sci. Pharm. 2010, 78, 1–12. [Google Scholar]
- Rotili, D.; Tarantino, D.; Artico, M.; Nawrozkij, M.B.; Gonzalez-Ortega, E.; Clotet, B.; Samuele, A.; Este, J.A.; Maga, G.; Mai, A. Diarylpyrimidine-dihydrobenzyloxopyrimidine hybrids: New, wide-spectrum anti-HIV-1 agents active at (sub)-nanomolar level. J. Med. Chem. 2010, 54, 3091–3096. [Google Scholar]
- Coumar, M.S.; Chu, C.Y.; Lin, C.W.; Shiao, H.Y.; Ho, Y.L.; Reddy, R.; Lin, W.H.; Chen, C.H.; Peng, Y.H.; Leou, J.S.; et al. Fast-forwarding hit to lead: Aurora and epidermal growth factor receptor kinase inhibitor lead identification. J. Med. Chem. 2010, 53, 4980–4988. [Google Scholar] [CrossRef]
- Saravanan, K.; Barlow, H.C.; Barton, M.; Calvert, A.H.; Golding, B.T.; Newell, D.R.; Northen, J.S.; Curtin, N.J.; Thomas, H.D.; Grin, R.J. Nucleoside transport inhibitors: Structure-activity relationships for pyrimido[5,4-d]pyrimidine derivatives that potentiate pemetrexed cytotoxicity in the presence of 1-acid glycoprotein. J. Med. Chem. 2011, 54, 1847–1859. [Google Scholar]
- Groth, D.E. Azoxystrobin rate and timing effects on rice head blast incidence and rice grain and milling yields. Plant Dis. 2006, 90, 1055–1058. [Google Scholar] [CrossRef]
- Rosenzweig, N.; Olaya, G.; Atallah, Z.K.; Cleere, S.; Stanger, C.; Stevenson, W.R. Monitoring and tracking changes in sensitivity to azoxystrobin fungicide in alternaria solani in Wisconsin. Plant Dis. 2008, 92, 555–560. [Google Scholar] [CrossRef]
- Heye, U.J.; Speich, J.; Siegle, H.; Steinemann, A.; Forster, B.; Knauf-Beiter, G.; Herzoga, J.; Hubele, A. CGA 219417: A novel broad-spectrum fungicide. Crop Prot. 1994, 13, 541–549. [Google Scholar] [CrossRef]
- Sholberg, P.L.; Bedford, K.; Stokes, S. Sensitivity of Penicillium spp. and Botrytis cinerea to pyrimethanil and its control of blue and gray mold of stored apples. Crop Prot. 2005, 24, 127–134. [Google Scholar] [CrossRef]
- Fustero, S.; Piera, J.; Sanz-Cervera, J.F.; Roman, R.; Brodsky, B.H.; Sanchez-Rosello, M.; Acen, J.L.; Arellano, C.R. New fluorinated 1,3-vinylogous amidines as versatile intermediates: Synthesis of fluorinated pyrimidin-2(1h)-ones. Tetrahedron 2006, 62, 1444–1451. [Google Scholar]
- Deng, X.Q.; Wei, C.X.; Li, F.N.; Sun, Z.G.; Quan, Z.S. Design and synthesis of 10-alkoxy-5, 6-dihydro-triazolo[4,3-d]benzo[f][1,4] oxazepine derivatives with anticonvulsant activity. Eur. J. Med. Chem. 2010, 45, 3080–3086. [Google Scholar]
- Achar, K.C.S.; Hosamani, K.M.; Seetharamareddy, H.R. In-vivo analgesic and anti-inflammatory activities of newly synthesized benzimidazole derivatives. Eur. J. Med. Chem. 2010, 45, 2048–2054. [Google Scholar] [CrossRef]
- Ding, Y.L.; Girardet, J.L.; Smith, K.L.; Larson, G.; Prigaro, Brett; Lai, V.C.H.; Zhong, W.; Wu, J.Z. Parallel synthesis of pteridine derivatives as potent inhibitors for hepatitis c virus ns5b rna-dependent rna polymerase. Bioorg. Med. Chem. Lett. 2005, 15, 675–678. [Google Scholar] [CrossRef]
- Qu, G.R.; Han, S.H.; Zhang, Z.G.; Geng, M.W.; Xue, F. Microwave assisted synthesis of 6-substituted aminopurine analogs in water. J. Braz. Chem. Soc. 2006, 17, 915–922. [Google Scholar] [CrossRef]
- Gangjee, A.; Zhao, Y.; Lin, L.; Raghavan, S.; Roberts, E.G.; Risinger, A.L.; Hamel, E.; Mooberry, S.L. Synthesis and discovery of water-soluble microtubule targeting agents that bind to the colchicine site on tubulin and circumvent pgp mediated resistance. J. Med. Chem. 2010, 53, 8116–8128. [Google Scholar] [CrossRef]
- Hu, Y.G.; Wang, Y.; Du, S.M.; Chen, X.B.; Ding, M.W. Efficient synthesis and biological evaluation of some 2,4-diamino-furo[2,3-d]pyrimidine derivatives. Bioorg. Med. Chem. Lett. 2010, 20, 6188–6190. [Google Scholar]
- Xu, H.; Jian, K.; Guan, Q.; Ye, F.; Lv, M. Antifungal activity of some diaryl ethers. Chem. Pharm. Bull. 2007, 55, 1755–1757. [Google Scholar] [CrossRef]
- Damodiran, M.; Muralidharan, D.; Perumal, P.T. Regioselective synthesis and biological evaluation of bis(indolyl)methane derivatized 1,4-disubstituted 1,2,3-bistriazoles as anti-infective agents. Bioorg. Med. Chem. Lett. 2009, 19, 3611–3514. [Google Scholar] [CrossRef]
- Fan, Z.J.; Shi, Z.G.; Zhang, H.K.; Liu, X.F.; Bao, L.L.; Ma, L.; Zuo, X.; Zheng, Q.X.; Mi, N. Synthesis and biological activity evaluation of 1,2,3-thiadiazole derivatives as potential elicitors with highly systemic acquired resistance. J. Agric. Food Chem. 2009, 57, 4279–4286. [Google Scholar]
- Caldiz, D.O.; Rolon, D.A.; Rico, J.D.; Andreu, A.B. Performance of dimethomorph + mancozeb applied to seed potatoes in early management of late blight(Phytophthora infestans). Potato Res. 2007, 50, 59–70. [Google Scholar] [CrossRef]
- Stein, J.M.; Kirk, W.W. Field optimization of dimethomorph for the control of potato late blight phytophthora infestans: Application rate, interval, and mixtures. Crop Prot. 2003, 22, 609–614. [Google Scholar]
- Banerjee, T.; Dureja, P. Antifungal activity of 4'-(2,6,6-trimethyl-2-cyclohexen-1-yl)-3'-butene- 2'-ketoxime n-o-alkyl ethers. J. Agric. Food Chem. 2011, in press.. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sun, L.; Wu, J.; Zhang, L.; Luo, M.; Sun, D. Synthesis and Antifungal Activities of Some Novel Pyrimidine Derivatives. Molecules 2011, 16, 5618-5628. https://doi.org/10.3390/molecules16075618
Sun L, Wu J, Zhang L, Luo M, Sun D. Synthesis and Antifungal Activities of Some Novel Pyrimidine Derivatives. Molecules. 2011; 16(7):5618-5628. https://doi.org/10.3390/molecules16075618
Chicago/Turabian StyleSun, Li, Jie Wu, Lingzi Zhang, Min Luo, and Dequn Sun. 2011. "Synthesis and Antifungal Activities of Some Novel Pyrimidine Derivatives" Molecules 16, no. 7: 5618-5628. https://doi.org/10.3390/molecules16075618




