Influence of Polarity of Solvents on the Spectral Properties of Bichromophoric Coumarins
Abstract
:1. Introduction
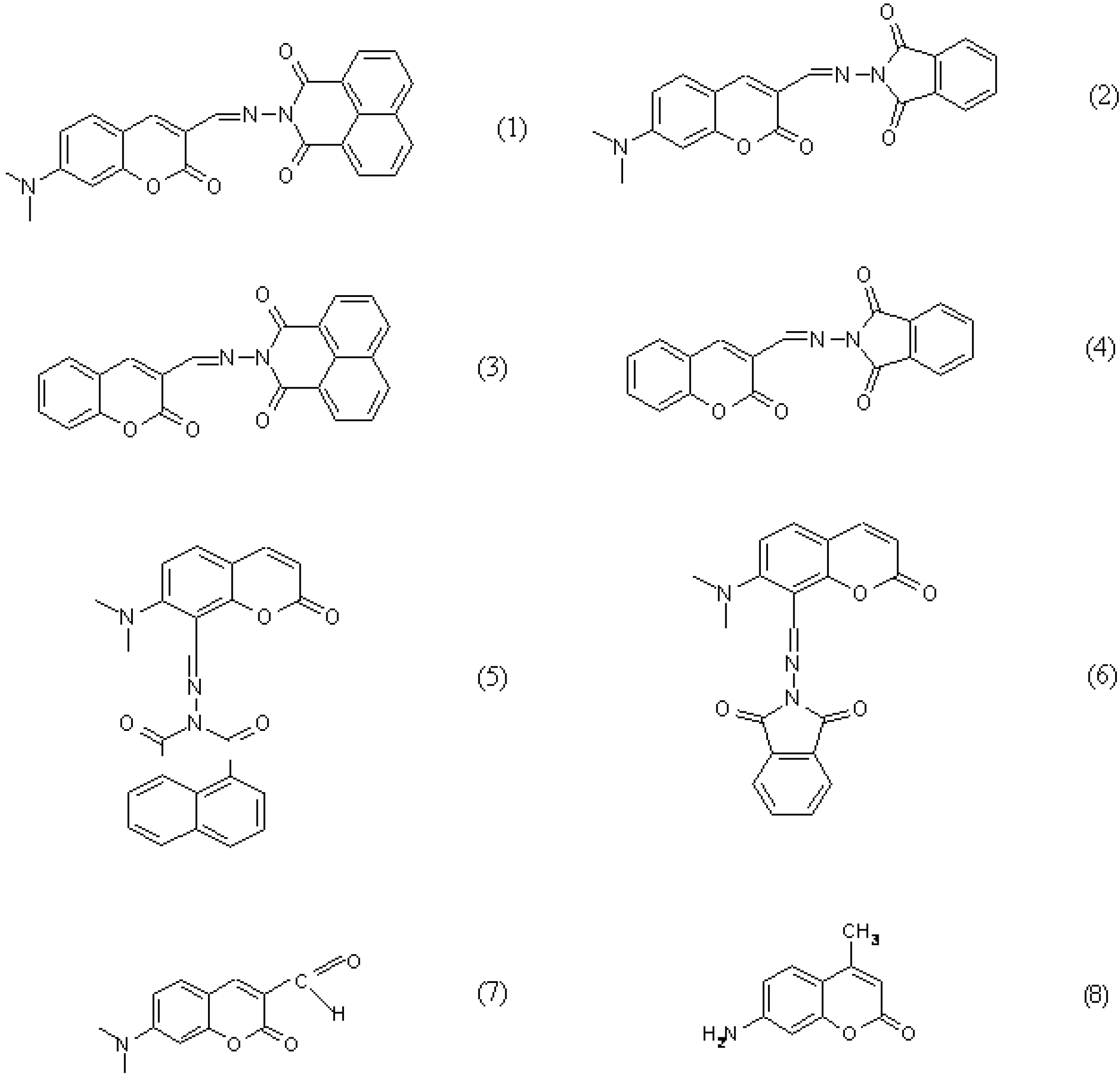
2. Results and Discussion
| Substrate a | chloroform | methanol | |||||||
|---|---|---|---|---|---|---|---|---|---|
| λA b nm | Logε c dm3mol−1 cm−1 | λF nm | Φr(A) e | τf ns | λA b nm | Logε c dm-3mol−1 cm−1 | λFd nm | Φr(A) e | |
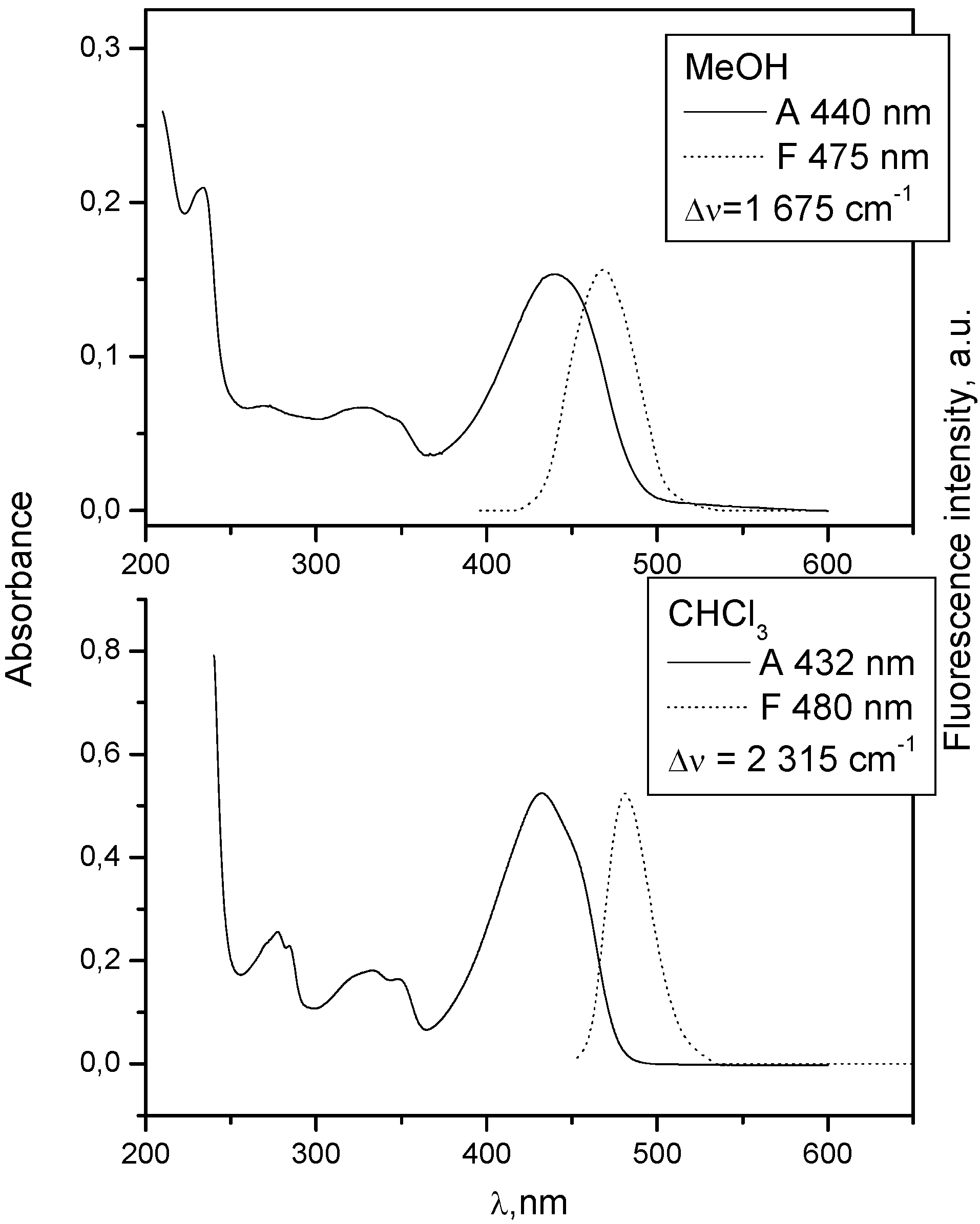
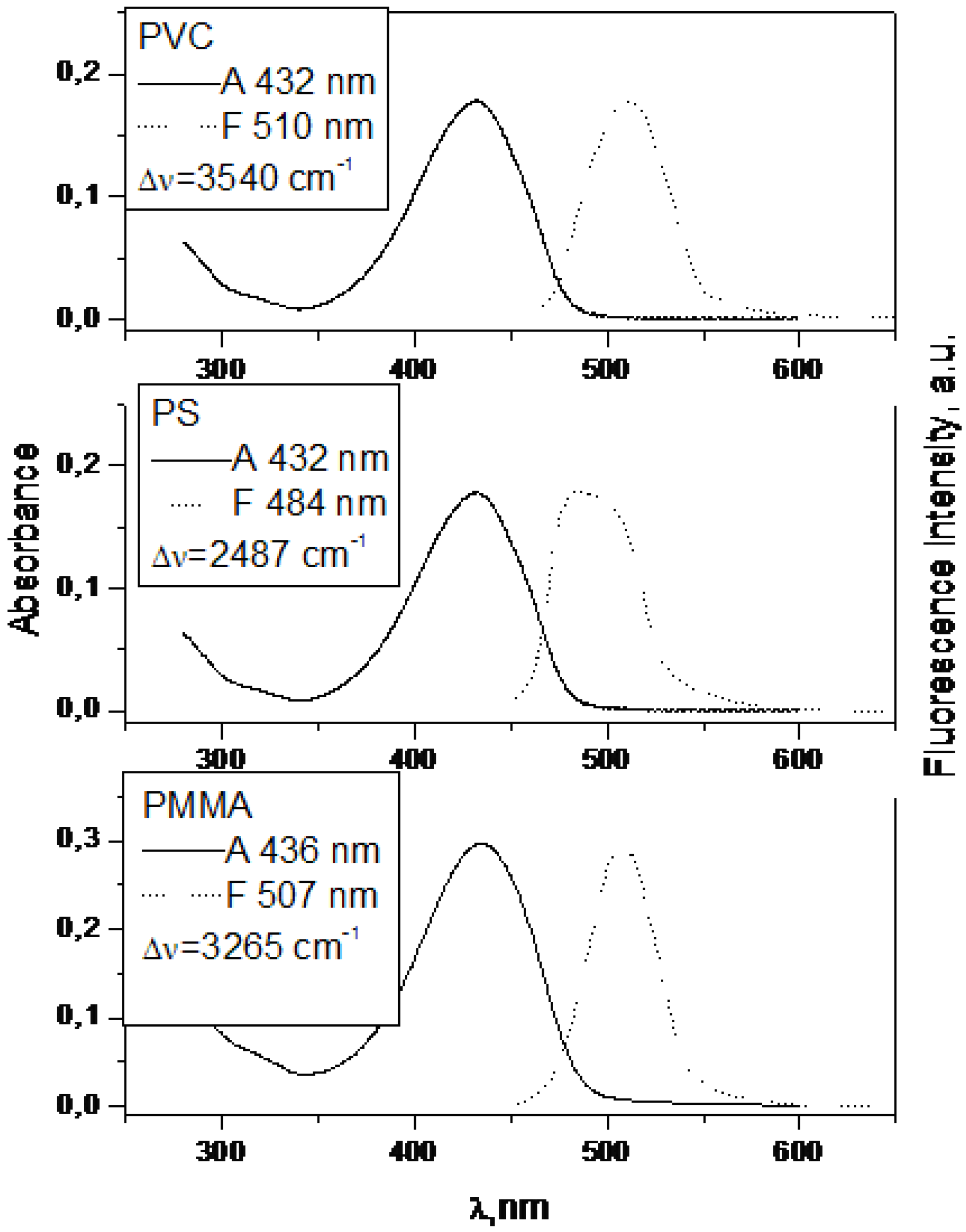
| 1 | 432 | 3.98 | 480 | 4.27 | 0.7 | 440 | 4.099 | 475 | 0.55 |
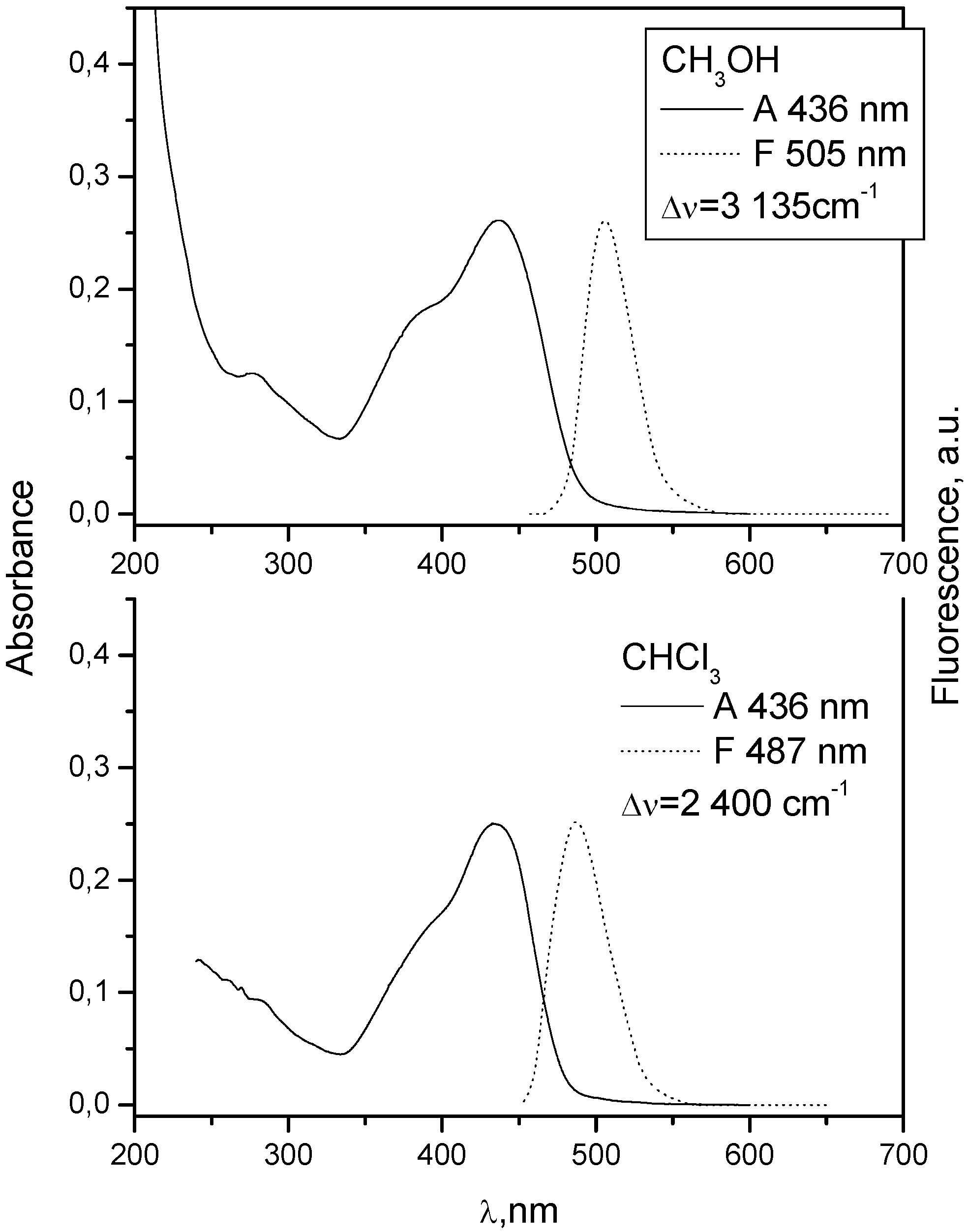
| 2 | 436 | 4.40 | 487 | 1.91 | 1.7 | 436 | 4.586 | 505 | 4.11 |
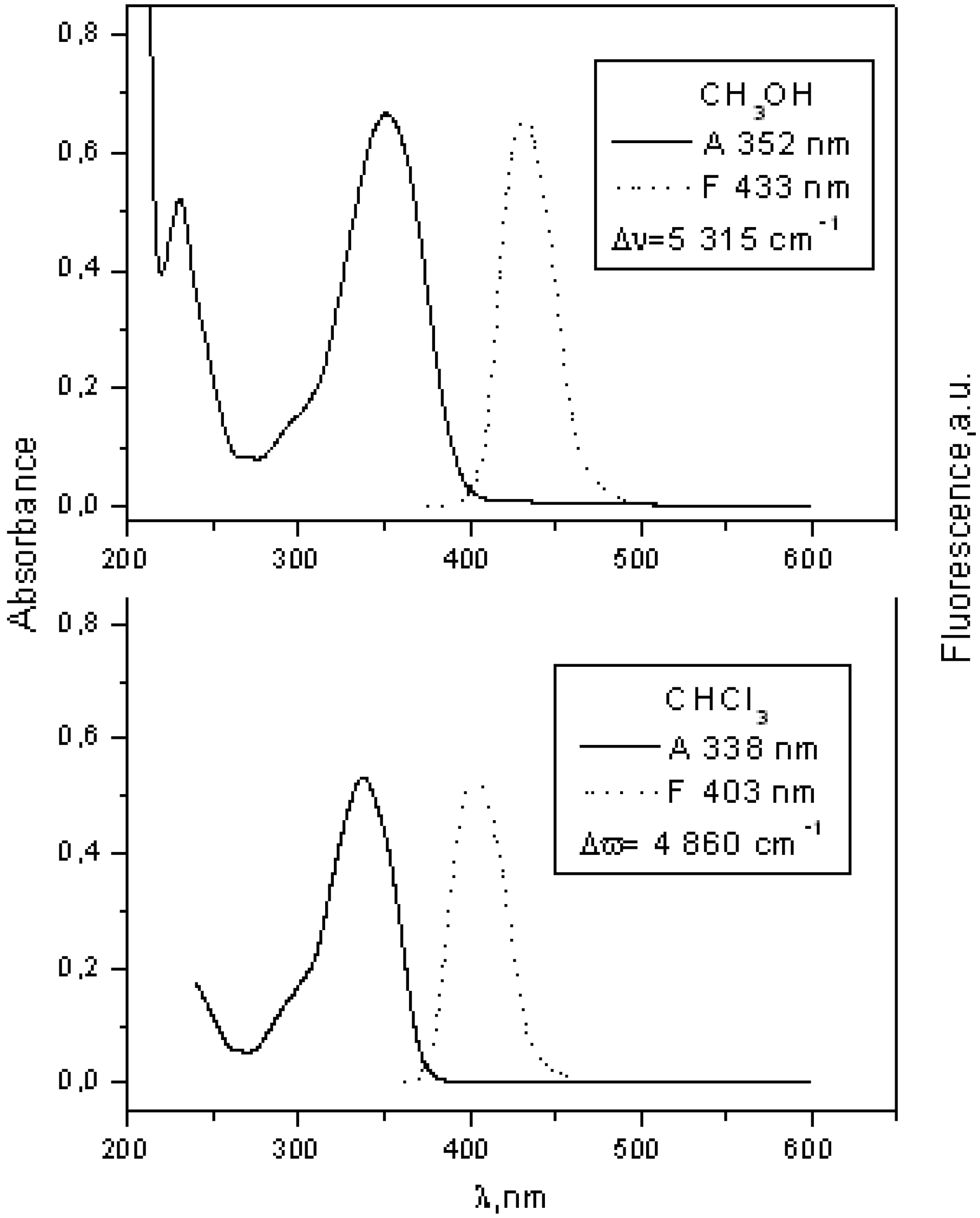
| 3 | 333 | 4.21 | 400 | 0.18 | 329 | 4.331 | 433 | 0.14 | |
| 4 | 356 | 4.59 | 405 | 0.05 | 352 | 4.352 | 438 | 0.09 | |
| 5 | 350 | 4.22 | 426 | 0.82 | 2.1 | 337 | 4.308 | 437 | 0.17 |
| 6 | 356 | 4.280 | 425 | 1.37 | 1.4 | 356 | 4.280 | 449 | 0.22 |
| 7 | 440 | 4.69 | 474 | 13.96 | 2.3 | 440 | 4.450 | 494 | 0.40 |
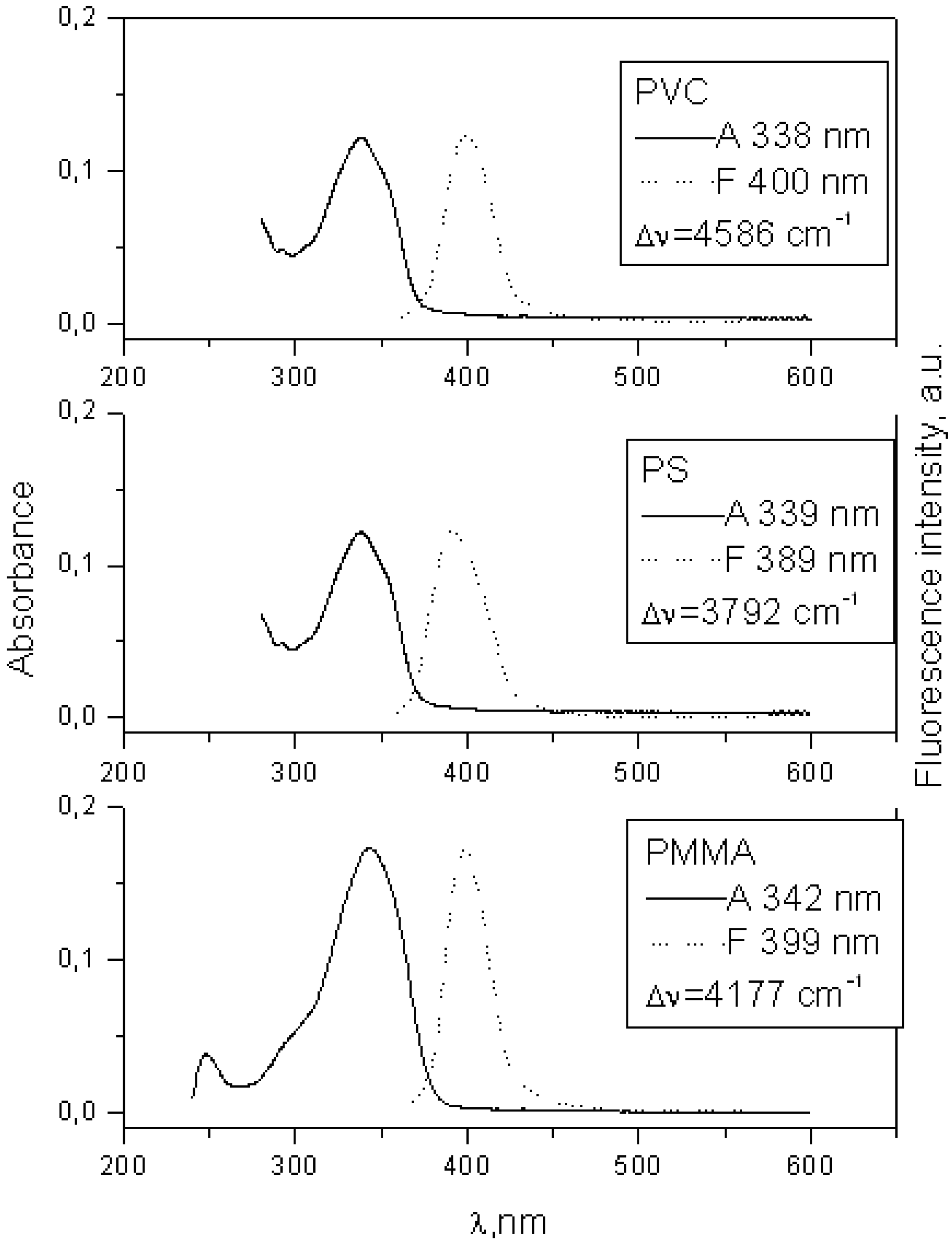
| 8 | 338 | 4.73 | 403 | 9.93 | 3.1 | 352 | 4.823 | 433 | 3.04 |
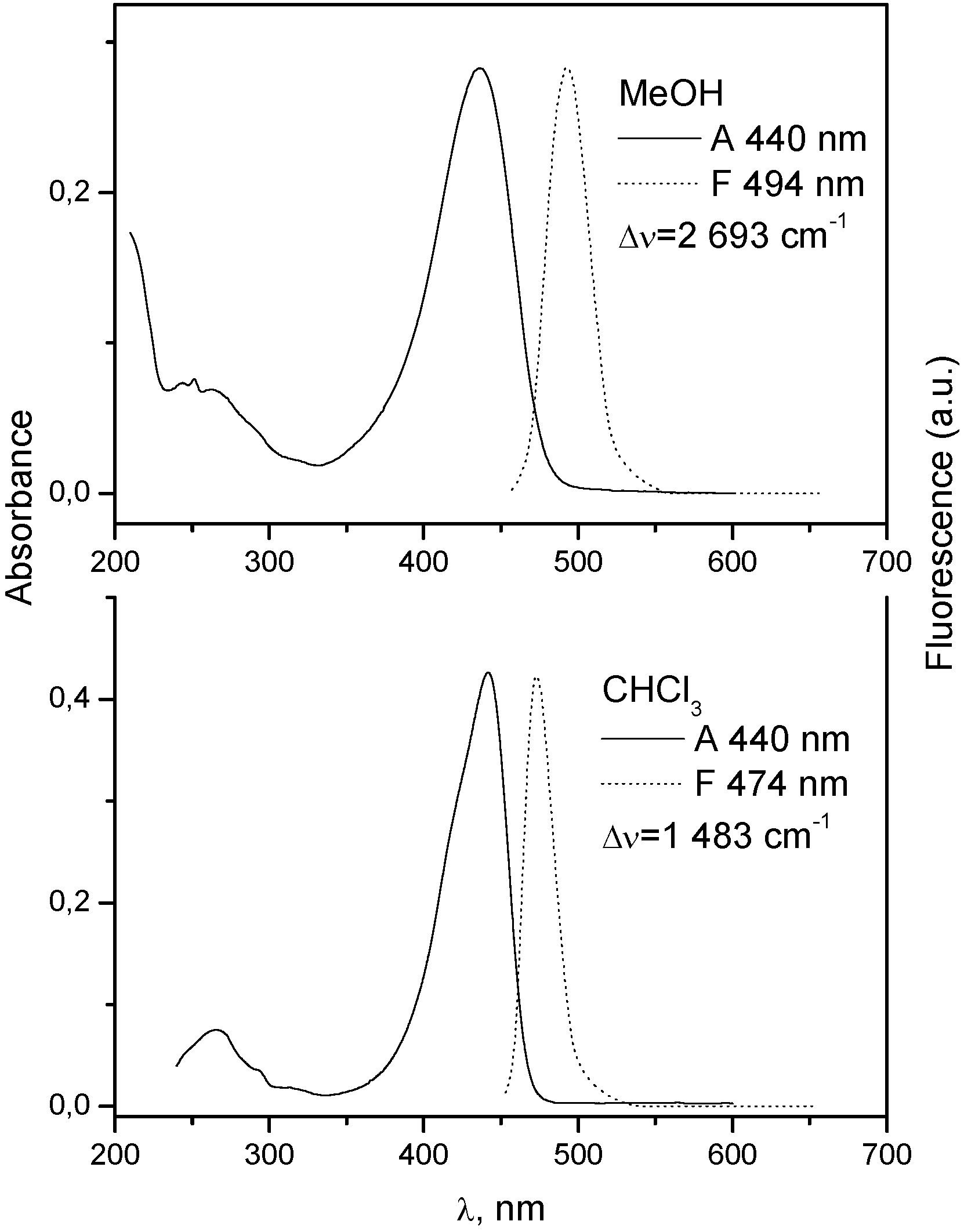
| Substrate a | PMMA | ||||
| λA b nm | λFc nm | Δυ d cm−1 | Φr(A) e | Τ f ns | |
| 1 | 432 | 474 | 2 050 | 9.2 | 3.1 |
| 2 | 435 | 507 | 3 265 | 8.6 | 2.7 |
| 3 | 333 | ||||
| 4 | 356 | 432 | 4 994 | 0.8 | 1.4 |
| 5 | 348 | 405 | 4 404 | 0.53 | 2.0 |
| 6 | 356 | 418 | 3 398 | 0.72 | 1.7 |
| 7 | 438 | 477 | 1 866 | 7.57 | 4.0 |
| 8 | 342 | 399 | 4 177 | 3.53 | 2.6 |
| Substrate a | PS | ||||
| λA b nm | λFc nm | Δυ d cm−1 | Φr(A) e | Τ f ns | |
| 1 | 433 | 461 | 1 403 | 3.7 | 1.5 |
| 2 | 432 | 484 | 2 487 | 3.4 | 1.9 |
| 3 | 334 | ||||
| 4 | 357 | 436 | 5 075 | 0.3 | 0.7 |
| 5 | 349 | 404 | 3 900 | 0.4 | 1.7 |
| 6 | 356 | 420 | 4 046 | 0.6 | 1.4 |
| 7 | 438 | 480 | 1 908 | 5.2 | 2.3 |
| 8 | 339 | 389 | 3 792 | 3.7 | 2.3 |
| Substrate a | PVC | ||||
| λA b nm | λFc nm | Δυ d cm−1 | Φr(A) e | Τ f ns | |
| 1 | 432 | 486 | 2 520 | 0.4 | 2.6 |
| 2 | 432 | 510 | 3 540 | 2.5 | 2.8 |
| 3 | 336 | ||||
| 4 | 357 | 436 | 5 075 | 0.1 | 1.6 |
| 5 | 350 | 404 | 3 820 | 0.1 | 2.8 |
| 6 | 359 | 423 | 4 214 | 0.1 | 2.3 |
| 7 | 445 | 480 | 1 639 | 1.0 | 4.3 |
| 8 | 338 | 400 | 4 586 | 0.92 | 1.9 |

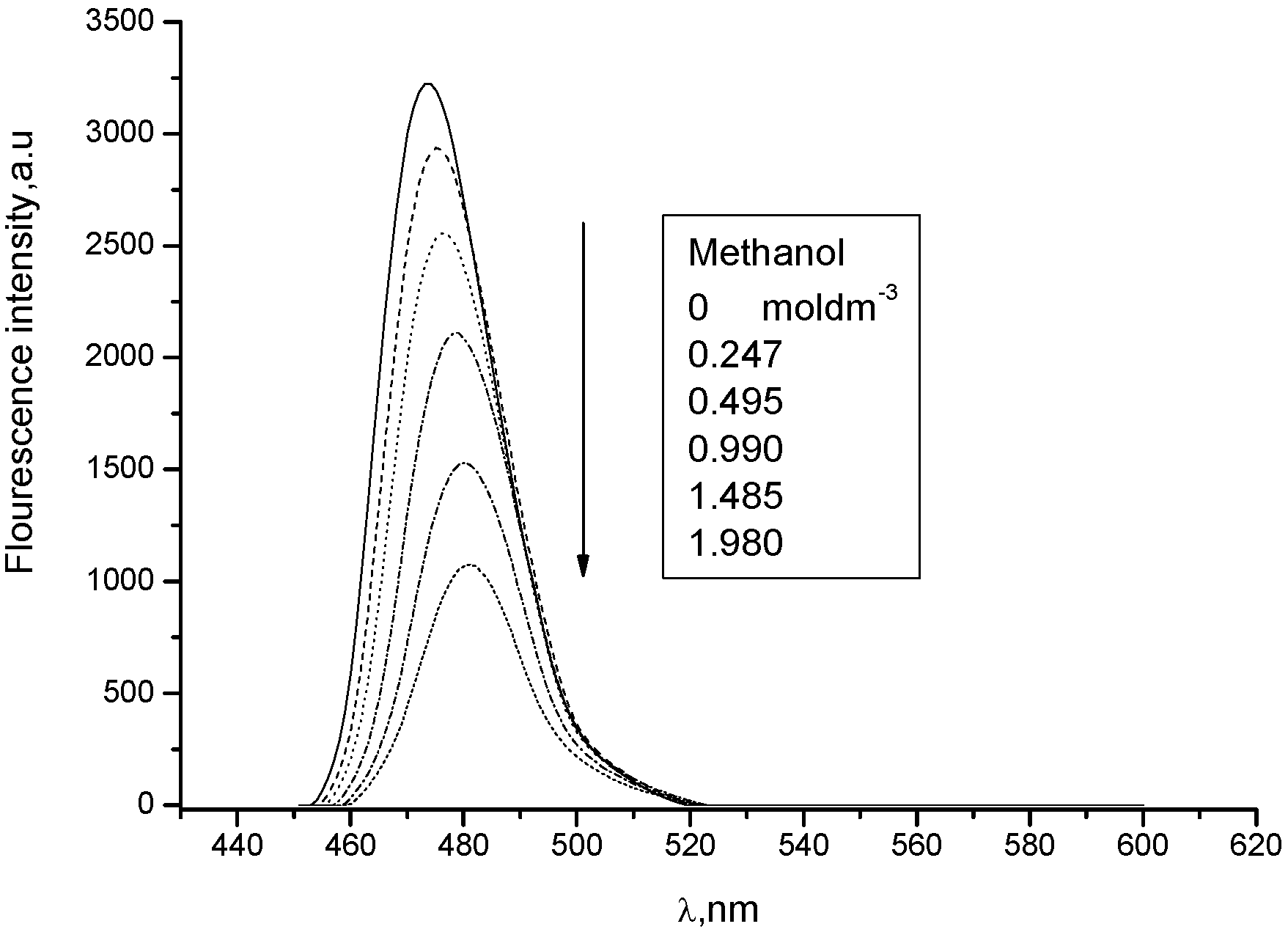
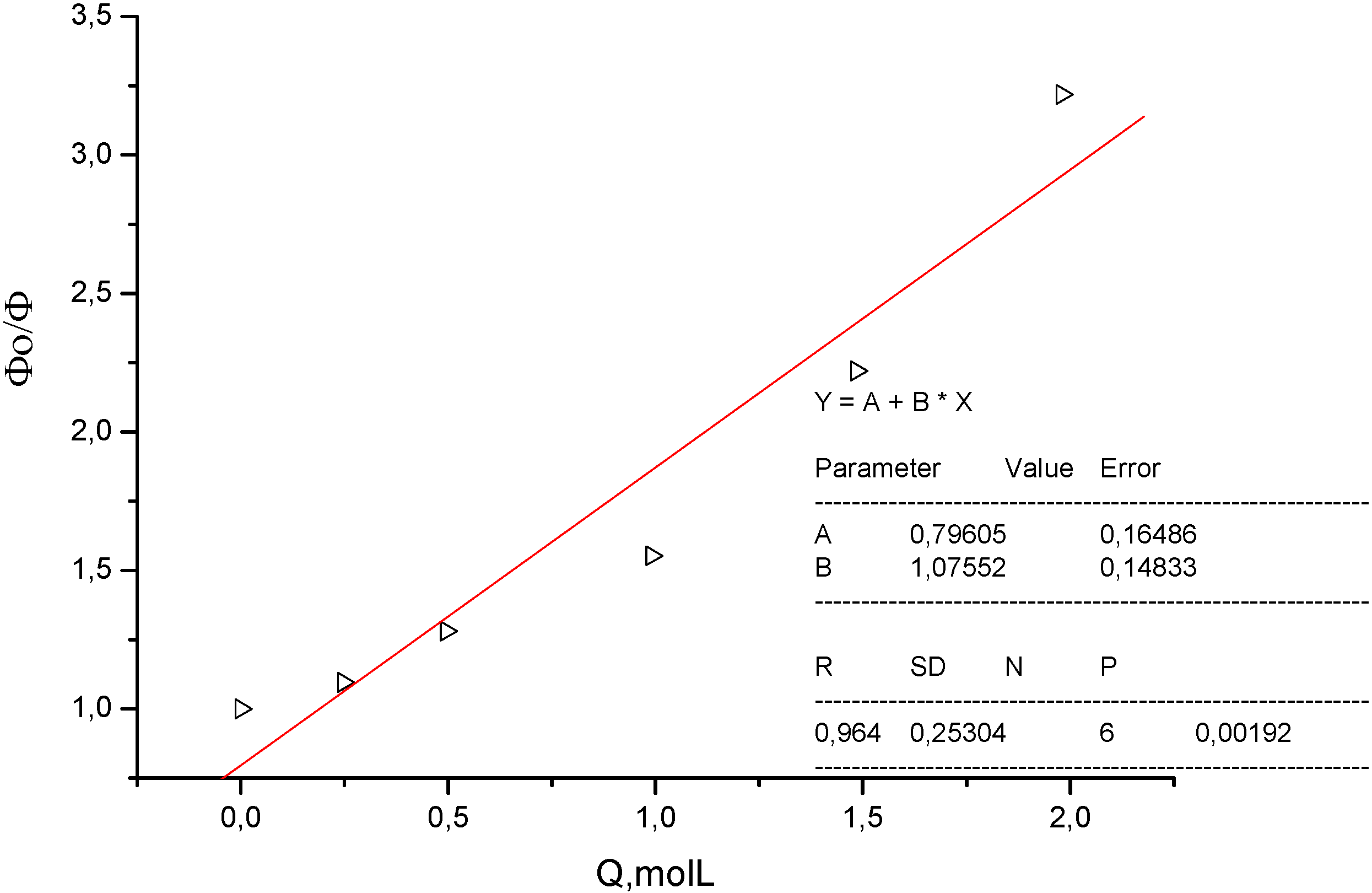
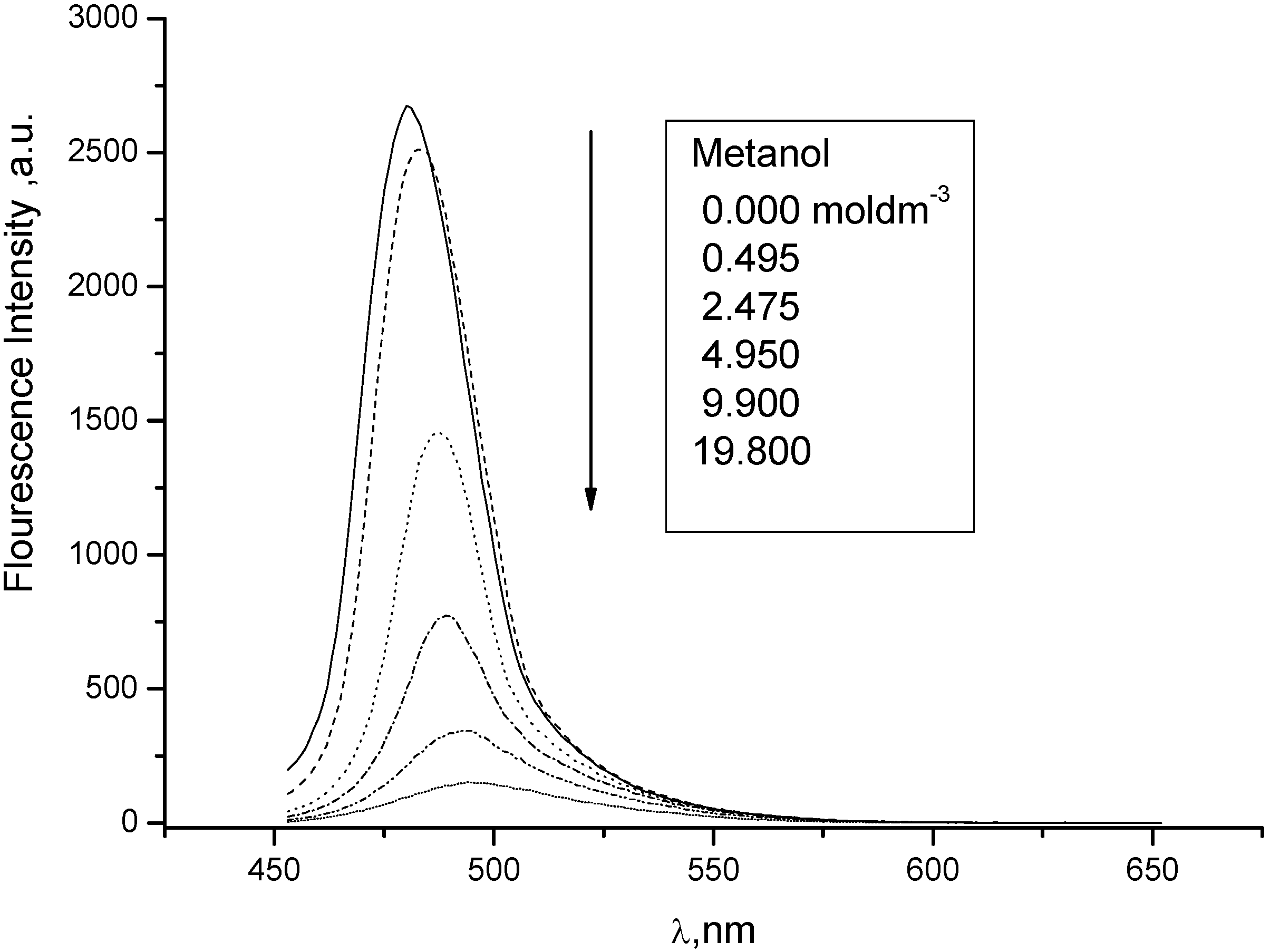
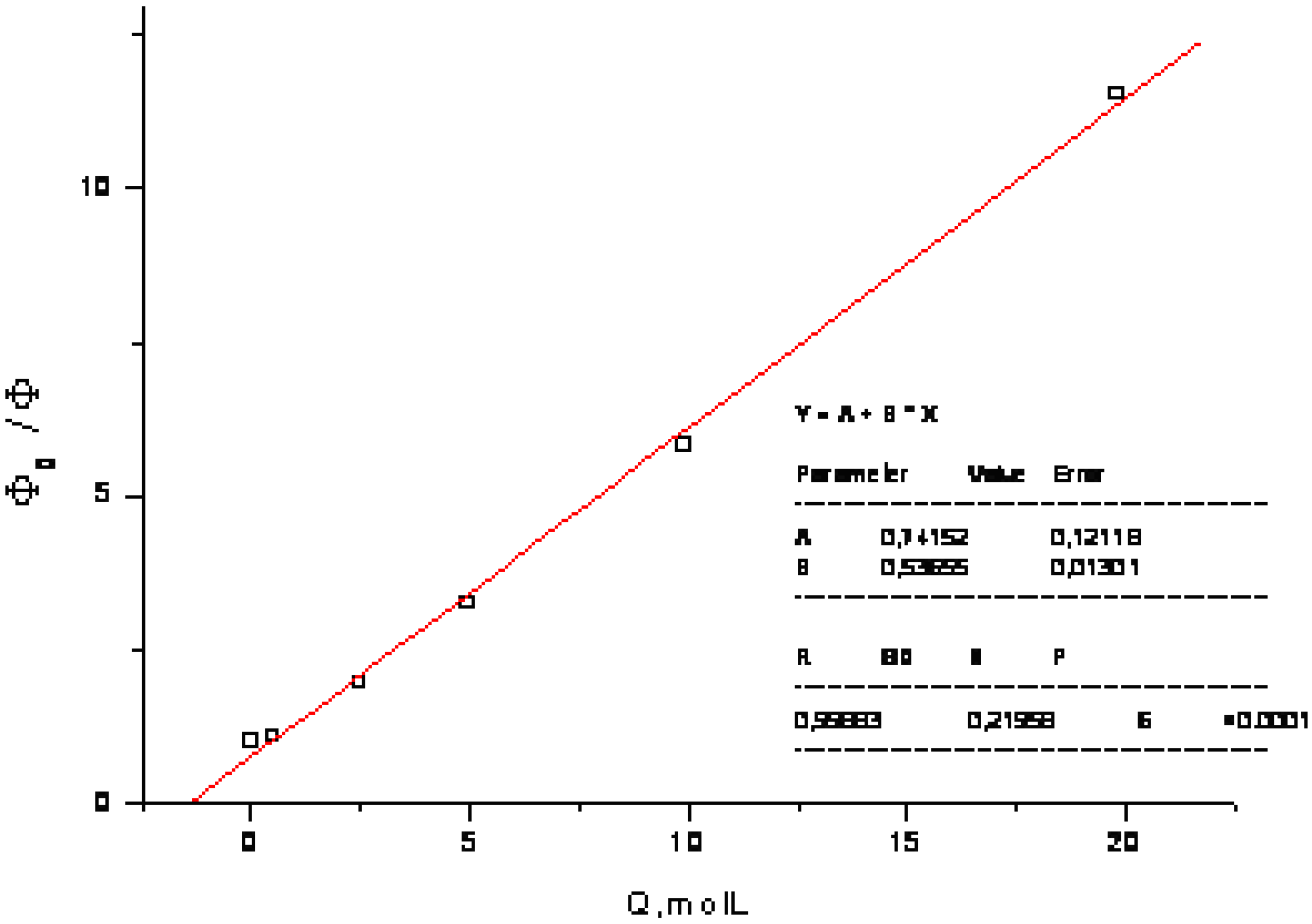
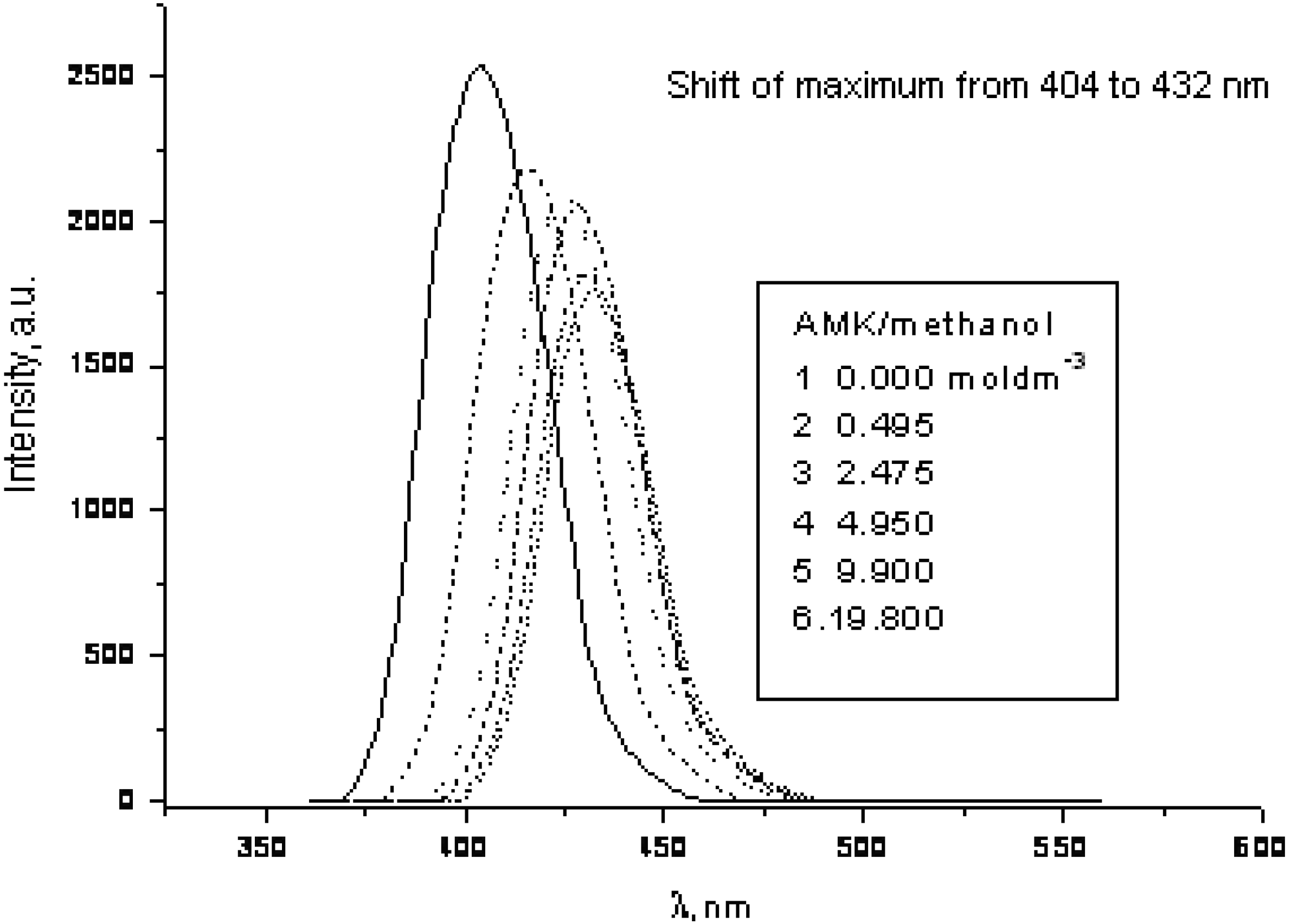

3. Experimental
3.1. Synthesis of Complex Coumarins
3.2. Spectral Measurements
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds are available from authors.
References and Notes
- Prasanna de Silva, A.; Gunaratante, H.Q.N.; Gunnlaugsson, Th.; Huxley, A.J.M.; Mc Coy, C.P.; Rademacher, J.T.; Rice, T.E. Signaling recognition events with fluorescent sensors and switches. Chem. Rev. 1997, 97, 1515–1566. [Google Scholar] [CrossRef]
- Prasanna de Silva, A.; Vance, T.P.; West, M.E.S.; Wright, G.D. Bright molecules with sense, logic, numeracy and utility. Org. Biomol. Chem. 2008, 6, 2468–2481. [Google Scholar] [CrossRef]
- Jones, G.; Jackson, W.R.; Choi, C.Y.; Bergman, W.R. Solvent effect on emission yield and lifetime for coumarin laser dyes. Requirement for rotatory decay mechanism. J. Phys. Chem. 1985, 89, 294–300. [Google Scholar] [CrossRef]
- Horng, M.L.; Gardeski, J.A.; Papazyan, A.; Maroncelli, M. Subpicosecond measurement of polar salvation dynamics: Coumarin 153 revisited. J. Phys. Chem. 1995, 99, 17311–17337. [Google Scholar]
- Arbeloa, T.L.; Arbeloa, F.L.; Arbeloa, I.L. Influence of fluorinated group on the photophysics of 7-aminocoumarins. J. Luminescence 1996, 68, 149–155. [Google Scholar] [CrossRef]
- Du, D.M.; Wang, Y.M.; Meng, J.B. Micro-environmental effects on the photochemical and photophysical processes of long chain coumarin esters. Chem. J. Chin. Univ.-Chin. 1998, 19, 1611–1613. [Google Scholar]
- Raju, B.B.; Costa, S.M.B. Photophysical properties of 7-diethylaminocoumarin dyes in dioxane-water mixtures: hydrogen bonding, dielectric enrichment and polarity effects. Phys. Chem. Chem. Phys. 1999, 1, 3539–3547. [Google Scholar]
- Ramalingam, A.; Sivaram, B.M.; Palamisamy, P.K.; Masilamani, V. Photophysics of TICT states of 7-diethylamino-4-methyl coumarin dye by energy transfer techniques. Spectrochim. Acta Part A-Molec. Biomolec. Spect. 2000, 56, 1205–1210. [Google Scholar] [CrossRef]
- Morimoto, A.; Yatsuhashi, T.; Shimada, T.; Biczok, L.; Tryk, D.A.; Inoue, H. Radiationless deactivation of an intramolecular charge transfer excited state through hydrogen bonding: effect of molecular structure and hard-soft anionic character in the excited state. J. Phys Chem. A 2001, 105, 10488–10496. [Google Scholar]
- Morlet-Savary, F.; Ley, C.; Jacques, P.; Fouasier, J.P. Photophysics of a bridged 7-diethylamino-4-methylcoumarin C102: Studying the hydrogen bonding effect by time resolved stimulated emission. J.Phys Chem. A 2001, 105, 11026–11033. [Google Scholar]
- Satpati, A.K.; Senthilkumar, S.; Kumbhakar, M.; Nath, S.; Maity, D.K.; Pal, H. Investigations of the solvent polarity effect on the photophysical properties of coumarin-7 dye. Photochem. Photobiol. 2005, 81, 270–278. [Google Scholar] [CrossRef]
- Satpati, A.K.; Kumbhakar, M.; Maity, D.K.; Pal, H. Photophysical investigations of the solvent polarity effect on properties of coumarin-6 dye. Chem Phys. Lett. 2005, 40i7, 114–118. [Google Scholar]
- Mandal, P.K.; Paul, A.; Samanta, A. Fluorescence studies of environmentally benign solvents: solvation dynamics of coumarin 102 in [BMIM][BF4]. Res. Chem. Intermed. 2005, 31, 575–583. [Google Scholar] [CrossRef]
- Raikar, U.S.; Renuka, C.G.; Nadaf, Y.F.; Mulimani, B.G.; Karguppikar, A.M. Rotational diffusion and solvatochromic correlation of coumarin 6 laser dye. J. Fluorescence 2006, 16, 847–854. [Google Scholar] [CrossRef]
- Raikar, U.S.; Renuka, C.G.; Nadaf, Y.F.; Mulimanim, B.G.; Karguppikar, A.M.; Soudagar, M.K. Solvent effects on the absorption and fluorescence spectra of coumarins 6 and 7 molecules: determination of ground state and excited state dipole moment. Spectrochim. Acta. Part A 2006, 65, 673–677. [Google Scholar] [CrossRef]
- Tablet, C.; Hildebrand, M. Quenching of the fluorescence of carboxy-5,6-benzocoumarin by aromatic amines. J. Photochem. Photobiol. A Chem. 2007, 189, 73–79. [Google Scholar] [CrossRef]
- Wang, T.; Zhao, Y.; Shi, M.; Wu, F. The synthesis of novel coumarin dyes and the study of their photoreaction properties. Dyes Pigments 2007, 75, 104–110. [Google Scholar] [CrossRef]
- Ammar, H.; Abid, S.; Fery-Forgues, S. Synthesis and spectroscopic study of new biscoumarin dyes based on 7-(4-methylcoumarinyl) diesters. Dyes Pigments 2008, 78, 1–7. [Google Scholar] [CrossRef]
- Winnik, F.M.; Regismond, S.T.A. Fluorescence methods in the study of the interactions of surfactants with polymers. Colloids Surfaces A: Physicochem. Engineer. Aspects 1996, 118, 1–36. [Google Scholar] [CrossRef]
- De Paula, R.; De Hora, A.E.; Machado De Miranda, J.A. 3-Benzoxazol-2-yl-7-(N,N-diethylamino)/chromen-2-one as a fluorescence probe for investigation of micellar microenvironments. J. Photochem. Photobiol. A: Chem. 2004, 165, 109–114. [Google Scholar] [CrossRef]
- Das, K.; Jain, B.; Gupta, P.K. Photophysics of coumarin 500 and coumarin 151 in AOT revers micelles. Chem. Phys. Lett. 2005, 410, 160–164. [Google Scholar] [CrossRef]
- Tamoto, Y.; Segawa, H.; Shirota, H. Solvation dynamics in aqueous anionic and cationic micelle solutions: sodium alkyl sulfate and alkyltrimethylamonium bromide. Langmuir 2005, 21, 3757–3764. [Google Scholar] [CrossRef]
- Scypinski, S.; Drake, J.M. Photophysics of coumarin inclusion complexes with inverted comples formation. J. Phys. Chem. 1985, 89, 2432–2435. [Google Scholar] [CrossRef]
- Chakraborty, A.; Seth, D.; Chakrabarty, D.; Sarkar, N. Photoinduced intermolecular electron transfer from aniline to 7-amino coumarin dyes in the surface of β-cyclodextrin. Spectrochim. Acta Part A 2006, 64, 801–808. [Google Scholar] [CrossRef]
- Li, D.; Zhang, J.; Anpo, M.; Xue, M.; Liu, Y. Photophysical properties of coumarin-6 molecules incorporated within MCM-48. Mater. Lett. 2005, 59, 2120–2123. [Google Scholar] [CrossRef]
- Guan, H.; Zhu, L.; Zhou, H.; Tang, H. Rapid probing of photoactivity on titania based self-cleaning materials using 7-hydroxycoumarin fluorescent probe. Anal. Chim. Acta 2008, 608, 73–78. [Google Scholar] [CrossRef]
- Takahashi, Y.; Shimada, R.; Maeda, A.; Kojima, K.; Uchida, K. Photophysics of coumarin 4 doped-amorphous silica glasses prepared by sol-gel method. J. Luminiscence 1996, 68, 187–192. [Google Scholar] [CrossRef]
- Stathatos, E.; Lianos, P.; Lavrencic-Stangar, U.; Orel, B. Study of laser action of coumarine-153 incorporated in sol-gel made silica/poly(propylene oxide) nanocomposite gels. Chem. Phys. Lett. 2001, 345, 381–385. [Google Scholar] [CrossRef]
- Takahashi, Y.; Maeda, A.; Kojima, K. Optical pH sensing characteristics in dye-doped sol-gel coating film base on the energy transfer. Jap. J. Appl. Phys Part 1 2003, 42, 4369–4377. [Google Scholar] [CrossRef]
- Yamaguchi, A.; Amino, Y.; Shima, K.; Suzuki, S.; Yamashita, T.; Teramae, N. Local environments of coumarin dyes within mesostructured silica-surfactant nanocomposites. J. Phys. Chem. B 2006, 110, 3910–3916. [Google Scholar] [CrossRef]
- Sabatini, C.A.; Pereira, R.V.; Gehlen, M.H. Fluorescence modulation of acridin and coumarine dyes by silver nanoparticles. J. Fluoescence 2007, 17, 377–382. [Google Scholar] [CrossRef]
- Soutar, I. The application of luminescence technique in polymer science. Polym. Int. 1991, 26, 35–49. [Google Scholar] [CrossRef]
- Morawetz, H. On the versatility of fluorescence technique in polymer research. J. Polym. Sci.: Part A: Polym. Chem. 1999, 37, 1725–1735. [Google Scholar] [CrossRef]
- Sarker, A.M.; Kaneko, Y.; Neckers, D.C. Photochemistry and photophysics of novel photoinitiators: N,N,N-tributyl-N-(4-methylene-7-methoxycoumarin)ammonium borates. J. Photochem. Photobiol. 1998, 117, 67–74. [Google Scholar] [CrossRef]
- Oh, J.K.; Wu, J.; Winnik, M.A.; Croun, G.P.; Rademacher, J.; Farwaha, R. Emulsion copolymerization of vinyl acetate and butylacetate in the presence of fluorescent dyes. J. Polym. Sci. Part A: Polym. Chem. 2002, 40, 1594–1604. [Google Scholar]
- Kaholek, M.; Hrdlovic, P. Spectral properties of coumarin derivatives substituted at position 3. Effect of polymer matrix. J. Photochem. Photobiol. A: Chem. 1997, 108, 283–288. [Google Scholar] [CrossRef]
- Kaholek, M.; Hrdlovic, P. Characteristics of excited states of 3-substituted coumarin derivatives and transfer of electronic energy to N-oxyl radicals. J. Photochem. Photobiol. A: Chem. 1999, 127, 45–55. [Google Scholar] [CrossRef]
- Kaholek, M.; Hrdlovic, P.; Bartos, J. Singlet probes based on coumarin derivatives substituted at position 3;spectral properties in solution and polymer matrices. Polymer 2000, 41, 991–1001. [Google Scholar] [CrossRef]
- Jones, G.; Jimenez, J.A.C. Azole-linked coumarin dyes as fluorescence probes of domain-forming polymers. J. Photochem. Photbiol. B: Biol. 2001, 65, 5–12. [Google Scholar] [CrossRef]
- Kim, C.; Trajkovskaja, A.; Wallace, J.U.; Chen, S.H. New insight into photoalignment of liquid crystals on coumarin containing polymer films. Macromolecules 2006, 39, 3817–3823. [Google Scholar] [CrossRef]
- Ley, J.G.E.; Tirumalasetty, P.P. Release chracteristics of polymethylmethcrytate nanospheres containing coumarin 6. J. Microencapsulation 2003, 20, 653–659. [Google Scholar]
- Mansour, A.F.; Killa, H.M.A.; El-Wanees, S.A.; El-Sayed, M.Y. Laser dyes doped with with (ST-co-MMA) as fluorescent solar collectors and their field performance. Polym. Testing 2005, 24, 519–525. [Google Scholar] [CrossRef]
- Felorzabihi, N.; Haley, J.C.; Bardajee, G.; Winnik, M.A. Systematic study of the fluorescence decay of amino-coumarins dyes in polymer matrices. J. Polym. Sci.: Part B: Polym. Phys. 2007, 45, 2333–2343. [Google Scholar] [CrossRef]
- Fu, Q.; Cheng, L.; Zhang, Y.; Shi, W. Preparation and reversible photocrosslinking/photo-cleavage of 4-methykcoumarin functionalized hyperbranched polyester. Polymer 2008, 49, 4981–4988. [Google Scholar] [CrossRef]
- Bayer, M.; Steger, D.; Fischer, K. The Luminescence of lignign-containing pulps – A comparison with fluorescence of model compounds in several media. J. Photochem. Photobiol. A: Chem. 1993, 76, 217–224. [Google Scholar] [CrossRef]
- Fischer, K.; Spange, S.; Fischer, S.; Bellmann, C.; Adams, J. Probing the surface polarity of native cellulose using genuine solvatochromic dyes. Cellulose 2002, 9, 31–40. [Google Scholar] [CrossRef]
- Coenjarts, C.; Garcia, O.; Llauger, L.; Vinette, A.L.; Scaiano, J.C. Mapping photogenerated radicals in thin polymer films: Fluorescence imaging using a prefluorescent radical probe. J. Am. Chem. Soc. 2003, 125, 620–621. [Google Scholar] [CrossRef]
- Scaiano, J.C.; Aliaga, C.; Chretien, M.N.; Frenette, M.; Focsaneanu, K.S.; Mikelson, L. Fluorescence sensor application as detector for DNA damage, free radical formation, and in microlithography. Pure Appl. Chem. 2005, 77, 1009–1018. [Google Scholar] [CrossRef]
- Viriota, M.L.; Carre, M.C.; Geoffroy-Chapotot, C.; Brembilla, A.; Muller, S.; Stoltz, J.F. Molecular rotors as fluorescent probes for biological studies. Clin. Hemorheol. Microcirculation 1998, 19, 151–160. [Google Scholar]
- Hrdlovic, P. Medium effect of polymer matrices on spectral properties of 4-aminophthalimide and 4-dimethylaminophthalimide. J. Macromol Sci. Part A Pure Appl. Chem. 2007, 44, 1047–1053. [Google Scholar] [CrossRef]
- Samanta, A.; Saroja, G. Steady state and time-resolved studies of the redox behaviour of 1,8-naphthalimide in the excited state. J. Photochem. Photobiol. A. Chem. 1994, 84, 19–26. [Google Scholar] [CrossRef]
- Hrdlovič, P.; Chmela, S.; Danko, M. Spectral characteristics and photochemical stability of fluorescence probes based on 1,8-naphthalimide in solution and in polymer matrix. J. Photochem. Photobiol. A. Chem. 1998, 112, 197–203. [Google Scholar] [CrossRef]
- Bojinov, V.; Ivanova, G.; Simeonov, D. Synthesis and photophysical investigations of novel polymerizable blue emitting fluorophores – combination of a hindered amine with a benzo[de]isoquinoline-1,3-dione. Macromol. Chem. Phys. 2004, 205, 1259–1268. [Google Scholar] [CrossRef]
- Gunnlaugasson, T.; Kruger, P.E.; Jansen, P.; Tierney, J.; Ali, H.D.P.; Hussey, G. Colorimetric ”Naked Eye” sensing of anions in aqueous solution. J. Org. Chem. 2005, 70, 10875–10878. [Google Scholar] [CrossRef]
- Abad, S.; Kluciar, M.; Miranda, M.A.; Pischel, U. Proton-induced fluorescence switching in novel naphthalimide-dansylamide dyads. J. Org. Chem. 2005, 70, 10565–10568. [Google Scholar] [CrossRef]
- Grabchev, I.; Chovelon, J.M.; Nedelcheva, A. Green fluorescence poly(amidoamine) dendrimer functionalized with 1,8-naphthalimide units as potential sensor for metal cations. J. Photochem. Photobiol. A. Chem. 2006, 183, 9–14. [Google Scholar] [CrossRef]
- Demets, G.J.F.; Triboni, E.R.; Alvarez, E.B.; Arantes, G.M.; Filho, P.B.; Politi, M.J. Solvent influence on the photophysical properties of 4-methoxy-N-methyl-1,8-naphthalimide. Spectrochim. Acta Part A 2006, 63, 220–226. [Google Scholar] [CrossRef]
- Magalhaes, J.L.; Pereira, R.V.; Triboni, E.R.; Berci Filho, P.; Gehlen, M.H.; Nart, F.C. Solvent influence on the photo-physical properties of 4-phenoxy-N-methyl-1,8-naphthalimide. Spectrochim. Acta Part A 2006, 183, 165–170. [Google Scholar]
- Prezhdo, O.V.; Uspenskii, B.V.; Prezhdo, V.V.; Boszczyk, W.; Distanov, V.B. Synthesis and spectral-luminescent characteristics 1,8-naphthalimides. Dyes Pigments 2007, 72, 42–46. [Google Scholar] [CrossRef]
- Bojinov, V.B.; Panova, I. Synthesis and absorption properties of new yellow-green emitting benzo[de]isoquinoiline-1,3-diones containing hindered amine and 2-hydroxyphenylbenzotriazole fragments. Dyes Pigments 2007, 74, 551–560. [Google Scholar] [CrossRef]
- Kollar, J.; Hrdlovic, P.; Chmela, S. Synthesis and spectral characteristics of di-substituted 1,8-naphthalimides; bi-radical formation. J. Photochem. Photobiol. A. Chem. 2008, 195, 64–71. [Google Scholar] [CrossRef]
- Kollar, J.; Hrdlovic, P.; Chmela, S. Synthesis and spectral characterics of substituted of 1,8-naphthalimides: intramolecular quenching by mono-nitroxides. J. Photochem. Photobiol. A. Chem. 2009, 204, 191–199. [Google Scholar] [CrossRef]
- Flašík, R.; Stankovičová, H.; Gáplovský, A.; Donovalová, J. Synthesis and study of novel derivatives potentially utilizable as memory media. Molecules 2009, 14, 4838–4848. [Google Scholar] [CrossRef]
- Kawski, A.; Kubicki, A.; Kuklinski, B.; Gryczynski, I. Unusual absorption and fluorescence properties of 1,6-diphenyl-1,3,5-hexatriene in poly(vinyl alcohol) film. J. Photochem. Photobiol. A: Chem. 1993, 71, 161–167. [Google Scholar] [CrossRef]
- Demas, J.N.; Adamson, A.W. Evaluation of photoluminescence lifetimes. J. Phys. Chem. 1971, 57, 2463–2466. [Google Scholar]
- Demas, J.N. Excited State Lifetime Measurements; Academic Press: New York, NY, USA, 1973; p. 245, Appendix E. [Google Scholar]
- Enderlein, J.; Erdmann, R. Fast fitting of multi-exponential decay curves. Opt. Commun. 1997, 134, 371–378. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hrdlovic, P.; Donovalova, J.; Stankovicova, H.; Gaplovsky, A. Influence of Polarity of Solvents on the Spectral Properties of Bichromophoric Coumarins. Molecules 2010, 15, 8915-8932. https://doi.org/10.3390/molecules15128915
Hrdlovic P, Donovalova J, Stankovicova H, Gaplovsky A. Influence of Polarity of Solvents on the Spectral Properties of Bichromophoric Coumarins. Molecules. 2010; 15(12):8915-8932. https://doi.org/10.3390/molecules15128915
Chicago/Turabian StyleHrdlovic, Pavol, Jana Donovalova, Henrieta Stankovicova, and Anton Gaplovsky. 2010. "Influence of Polarity of Solvents on the Spectral Properties of Bichromophoric Coumarins" Molecules 15, no. 12: 8915-8932. https://doi.org/10.3390/molecules15128915




