Palladium(II)/Cationic 2,2’-Bipyridyl System as a Highly Efficient and Reusable Catalyst for the Mizoroki-Heck Reaction in Water
Abstract
:1. Introduction
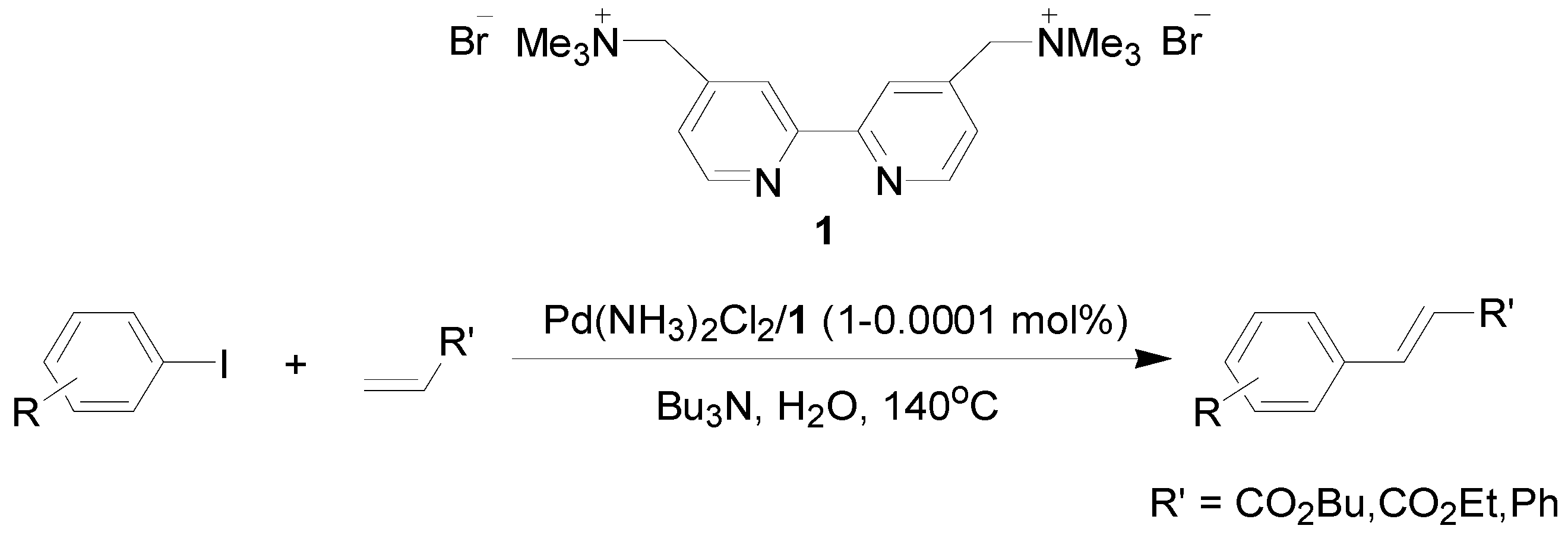
2. Results and Discussion
| Entry | Base (equiv.) | Yield (%)b |
|---|---|---|
| 1 | NaOH (2) | NR |
| 2 | NaHCO3 (2) | 2 |
| 3 | KOH (2) | NR |
| 4 | KOAc (2) | NR |
| 5 | K2CO3 (2) | NR |
| 6 | KF (2) | 4 |
| 7 | Et3N (2) | 32 |
| 8 | Diisopropylamine (2) | 17 |
| 9 | N,N-Diisopropylethylamine (2) | 6 |
| 10 | Bu3N (2) | 99 |
| 11 | Bu3N (1) | 49 |
| 12c | Bu3N (2) | NR |
| 13d | Bu3N (2) | 34 |
| Entry | Aryl iodide | Pd/1 (mol %) | Time (h) | Product (%)b | TON | ||
|---|---|---|---|---|---|---|---|
| 1c |  | 2a | 0.0001 | 48 |  | 4a (48) | 480,000 |
| 2 |  | 2b | 0.01 | 12 |  | 4b (99) | 9,900 |
| 3c | 2b | 0.0001 | 48 | 4b (92) | 920,000 | ||
| 4 |  | 2c | 0.01 | 12 |  | 4c (99) | 9,900 |
| 5c | 2c | 0.0001 | 48 | 4c (80) | 800,000 | ||
| 6 |  | 2d | 0.01 | 12 |  | 4d (99) | 9,900 |
| 7c | 2d | 0.0001 | 48 | 4d (86) | 860,000 | ||
| 8 |  | 2e | 0.01 | 12 |  | 4e (91) | 9,100 |
| 9c | 2e | 0.0001 | 48 | 4e (68) | 680,000 | ||
| 10 |  | 2f | 0.01 | 12 |  | 4f (92) | 9,200 |
| 11c | 2f | 0.0001 | 48 | 4f (91) | 910,000 | ||
| 12 |  | 2g | 0.01 | 12 |  | 4g (67) | 6,700 |
| 13 | 2g | 0.01 | 48 | 4g (94) | 9,400 | ||
| 14 |  | 2h | 0.01 | 48 |  | 4h (92) | 9,200 |
| 15 |  | 2i | 0.01 | 48 |  | 4i (56) | 5,600 |
| 16 |  | 2j | 0.01 | 48 |  | 4j (96) | 9,600 |
| 17 |  | 2k | 0.01 | 48 |  | 4k (95) | 9,500 |
| 18 |  | 2l | 0.01 | 48 |  | 4l (96) | 9,600 |
| Entry | Aryl iodide | Pd/1 (mol %) | Time (h) | Product (%)b | TON | ||
|---|---|---|---|---|---|---|---|
| 1 |  | 2a | 0.01 | 12 |  | 5a (63) | 6,300 |
| 2c | 2a | 0.0001 | 48 | 5a (58) | 580,000 | ||
| 3 |  | 2b | 0.01 | 12 |  | 5b (82) | 8,200 |
| 4c | 2b | 0.0001 | 48 | 5b (81) | 810,000 | ||
| 5 |  | 2c | 0.01 | 12 |  | 5c (74) | 7,400 |
| 6c | 2c | 0.0001 | 48 | 5c (51) | 510,000 | ||
| 7 |  | 2d | 0.01 | 12 |  | 5d (80) | 8,000 |
| 8c | 2d | 0.0001 | 48 | 5d (76) | 760,000 | ||
| 9 |  | 2e | 0.01 | 12 |  | 5e (81) | 8,100 |
| 10c | 2e | 0.0001 | 48 | 5e (45) | 450,000 | ||
| 11 |  | 2f | 0.01 | 12 |  | 5f (82) | 8,200 |
| 12c | 2f | 0.0001 | 48 | 5f (81) | 810,000 | ||
| 13 |  | 2g | 0.01 | 48 |  | 5g (82) | 8,200 |
| 14 |  | 2h | 0.01 | 48 |  | 5h (74) | 7,400 |
| 15 |  | 2i | 0.01 | 48 |  | 5i (44) | 4,400 |
| 16 |  | 2j | 0.01 | 48 |  | 5j (59) | 5,900 |
| 17 |  | 2k | 0.01 | 48 |  | 5k (65) | 6,500 |
| Entry | Aryl iodide | Pd/1 (mol %) | TBAB (equiv) | Product (%)b | ||
|---|---|---|---|---|---|---|
| 1 |  | 2a | 1 | 0 |  | 6a (36) |
| 2 | 2a | 1 | 1 | 6a (89) | ||
| 3 |  | 2b | 1 | 1 |  | 6b (93) |
| 4 |  | 2c | 1 | 1 |  | 6c (82) |
| 5 |  | 2d | 1 | 1 |  | 6d (83) |
| 6 |  | 2e | 1 | 1 |  | 6e (76) |
| 7 |  | 2f | 1 | 1 |  | 6f (87) |
| 8 |  | 2g | 1 | 1 |  | 6g (82) |
| 9 |  | 2h | 1 | 1 |  | 6h (71) |
| 10 |  | 2i | 1 | 1 |  | 6i (69) |
| 11 |  | 2j | 1 | 1 |  | 6j (68) |
| 12 |  | 2k | 1 | 1 |  | 6k (73) |
| 13 |  | 2l | 1 | 1 |  | 6l (93) |
| 14 |  | 2m | 1 | 1 |  | 6m (84) |

| Entry | Alkene | Time (h) | Pd/1 (mol %) | Cycle (%)b | |||||
|---|---|---|---|---|---|---|---|---|---|
| 1st | 2nd | 3rd | 4th | 5th | |||||
| 1 |  | 3a | 12 | 0.01 | 99 | 98 | 95 | 93 | 90 |
| 2 |  | 3b | 12 | 0.01 | 90 | 90 | 86 | 83 | 81 |
| 3c |  | 3c | 24 | 1 | 93 | 94 | 93 | 92 | 90 |
3. Experimental
3.1. General
3.2. Typical procedure for the Mizoroki-Heck reaction
3.3. Typical procedure for the reuse of the catalytic aqueous solution
4. Conclusions
Acknowledgements
References
- Mizoroki, T.; Mori, K.; Ozaki, A. Arylation of olefin with aryl iodide catalyzed by palladium. Bull. Chem. Soc. Jpn. 1971, 44, 581–581. [Google Scholar] [CrossRef]
- Heck, R.F.; Nolley, J.P., Jr. Palladium-Catalyzed vinylic hydrogen substitution reactions with aryl, benzyl, and styryl halides. J. Org. Chem. 1972, 37, 2320–2322. [Google Scholar] [CrossRef]
- Nicolaou, K.C.; Bulger, P.G.; Sarlah, D. Palladium-catalyzed cross-coupling reactions in total synthesis. Angew. Chem. Int. Ed. 2005, 44, 4442–4489. [Google Scholar] [CrossRef]
- Dounay, A.B.; Overman, L.E. The asymmetric intramolecular Heck reaction in natural product total synthesis. Chem. Rev. 2003, 103, 2945–2964. [Google Scholar] [CrossRef]
- Whitcombe, N.J.; Hii, K.K.; Gibson, S.E. Advances in the Heck chemistry of aryl bromides and chlorides. Tetrahedron 2001, 57, 7449–7476. [Google Scholar] [CrossRef]
- Beletskaya, I.P.; Cheprakov, A.V. The Heck reaction as a sharpening stone of palladium catalysis. Chem. Rev. 2000, 100, 3009–3066. [Google Scholar] [CrossRef]
- Shaughnessy, K.H. Hydrophilic ligands and their application in aqueous-phase metal-catalyzed reactions. Chem. Rev. 2009, 109, 643–710. [Google Scholar] [CrossRef]
- Horváth, I.T. Solvents from nature. Green Chem. 2008, 10, 1024–1028. [Google Scholar] [CrossRef]
- Li, C.J.; Trost, B.M. Green chemistry for chemical synthesis. Proc. Natl. Acad. Sci. USA 2008, 105, 13197–13202. [Google Scholar] [CrossRef]
- Liu, S.; Xiao, J. Toward green catalytic synthesis—Transition metal-catalyzedreactions in non-conventional media. J. Mol. Catal. A Chem. 2007, 270, 1–43. [Google Scholar] [CrossRef]
- Chen, L.; Li, C.J. Catalyzed reactions of alkynes in water. Adv. Synth. Catal. 2006, 348, 1459–1484. [Google Scholar] [CrossRef]
- Li, C.J.; Chen, L. Organic chemistry in water. Chem. Soc. Rev. 2006, 35, 68–82. [Google Scholar]
- Sheldon, R.A. Green solvents for sustainable organic synthesis: State of the art. Green Chem. 2005, 7, 267–278. [Google Scholar] [CrossRef]
- Li, C.J. Organic Reactions in aqueous media with a focus on carbon−carbon bond formations: A decade update. Chem. Rev. 2005, 105, 3095–3166. [Google Scholar] [CrossRef]
- Jeffery, T. On the efficiency of tetraalkylammonium salts in Heck type reactions. Tetrahedron 1996, 52, 10113–10130. [Google Scholar] [CrossRef]
- Arvela, R.K.; Leadbeater, N.E. Microwave-promoted Heck coupling using ultralow metal catalyst concentrations. J. Org. Chem. 2005, 70, 1786–1790. [Google Scholar] [CrossRef]
- Solabannavar, S.B.; Desai, U.V.; Mane, R.B. Heck reaction in aqueous medium using Amberlite IRA-400 (basic). Green Chem. 2002, 4, 347–348. [Google Scholar] [CrossRef]
- Kumar, N.S.C.R.; Raj, I.V.P.; Sudalai, A. Sulfonamide- and hydrazine-based palladium catalysts: Stable and efficient catalysts for C-C coupling reactions in aqueous medium. J. Mol. Catal. A Chem. 2007, 269, 218–224. [Google Scholar] [CrossRef]
- Dawood, K.M. Microwave-assisted Suzuki-Miyaura and Heck-Mizoroki cross-coupling reactions of aryl chlorides and bromides in water using stable benzothiazole-based palladium(II) precatalysts. Tetrahedron 2007, 63, 9642–9651. [Google Scholar] [CrossRef]
- Uozumi, Y.; Watanabe, T. Green catalysis: hydroxycarbonylation of aryl halides in water catalyzed by an amphiphilic resin-supported phosphine-palladium complex. J. Org. Chem. 1999, 64, 6921–6923. [Google Scholar] [CrossRef]
- Arvela, R.K.; Pasquini, S.; Larhed, M. Highly regioselective internal Heck arylation of hydroxyalkyl vinyl ethers by aryl halides in water. J. Org. Chem. 2007, 72, 6390–6396. [Google Scholar] [CrossRef]
- Botella, L.; Nájera, C. Mono- and β,β-double-Heck reactions of α,β-unsaturated carbonyl compounds in aqueous media. J. Org. Chem. 2005, 70, 4360–4369. [Google Scholar] [CrossRef]
- Gülcemal, S.; Kahraman, S.; Daran, J.C.; Çetinkaya, E.; Çetinkaya, B. The synthesis of oligoether-substituted benzimidazolium bromides and their use as ligand precursors for the Pd-catalyzed Heck coupling in water. J. Organomet. Chem. 2009, 694, 3580–3589. [Google Scholar] [CrossRef]
- Pawar, S.S.; Dekhane, D.V.; Shingare, M.S.; Thore, S.N. Sodium 2-(2-pyridin-3-ylethylamino)sulfonate: An efficient ligand and base for palladium-catalyzed Heck reaction in aqueous media. Tetrahedron Lett. 2008, 49, 4252–4255. [Google Scholar]
- Mukhopadhyay, S.; Rothenberg, G.; Joshi, A.; Baidossi, M.; Sasson, Y. Heterogeneous palladium-catalysed Heck reaction of aryl chlorides and styrene in water under mild conditions. Adv. Synth. Catal. 2002, 344, 348–354. [Google Scholar] [CrossRef]
- Hagiwara, H.; Sugawara, Y.; Hoshi, T.; Suzuki, T. Sustainable Mizoroki-Heck reaction in water: Remarkably high activity of Pd(OAc)2 immobilized on reversed phase silica gel with the aid of an ionic liquid. Chem. Commun. 2005, 2942–2944. [Google Scholar]
- Schönfelder, D.; Nuyken, O.; Weberskirch, R. Heck and Suzuki coupling reactions in water using poly(2-oxazoline)s functionalized with palladium carbene complexes as soluble, amphiphilic polymer supports. J. Organomet. Chem. 2005, 690, 4648–4655. [Google Scholar] [CrossRef]
- Zheng, P.; Zhang, W. Synthesis of efficient and reusable palladium catalyst supported on pH-responsive colloid and its application to Suzuki and Heck reaction in water. J. Catal. 2007, 250, 324–330. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, L.; Cui, Y. Catalytic performance of cellulose supported palladium complex for Heck reaction in water. J. Appl. Polym. Sci. 2008, 110, 2996–3000. [Google Scholar] [CrossRef]
- Wan, Y.; Wang, H.; Zhao, Q.; Klingstedt, M.; Terasaki, O.; Zhao, D. Ordered mesoporous Pd/silica-carbon as a highly active heterogeneous catalyst for coupling reaction of chlorobenzene in aqueous media. J. Am. Chem. Soc. 2009, 131, 4541–4550. [Google Scholar] [CrossRef]
- Firouzabadi, H.; Iranpoor, N.; Gholinejad, M. 2-Aminophenyl diphenylphosphinite as a new ligand for heterogeneous palladium-catalyzed Heck-Mizoroki reactions in water in the absence of any organic co-solvent. Tetrahedron 2009, 65, 7079–7084. [Google Scholar] [CrossRef]
- Qiao, K.; Sugimura, R.; Bao, Q.; Tomida, D.; Yokoyama, C. An efficient Heck reaction in water catalyzed by palladium nanoparticles immobilized on imidazolium-styrene copolymers. Catal. Commun. 2008, 9, 2470–2474. [Google Scholar] [CrossRef]
- Senra, J.D.; Malta, L.F.B.; da Costa, M.E.H.M.; Michel, R.C.; Aguiar, L.C.S.; Simas, A.B.C.; Antunes, O.A.C. Hydroxypropyl-α- cyclodextrin-capped palladium nanoparticles: active scaffolds for efficient carbon-carbon bond forming cross-couplings in water. Adv. Synth. Catal. 2009, 351, 2411–2422. [Google Scholar] [CrossRef]
- Sawoo, S.; Srimani, D.; Dutta, P.; Lahiri, R.; Sarkar, A. Size controlled synthesis of Pd nanoparticles in water and their catalytic application in C–C coupling reactions. Tetrahedron 2009, 65, 4367–4374. [Google Scholar] [CrossRef]
- Wu, W.Y.; Chen, S.N.; Tsai, F.Y. Recyclable and highly active cationic 2,2’-bipyridyl palladium(II) catalyst for Suzuki cross-coupling reaction in water. Tetrahedron Lett. 2006, 47, 9267–9270. [Google Scholar] [CrossRef]
- Chen, S.N.; Wu, W.Y.; Tsai, F.Y. Hiyama reaction of aryl bromides with arylsiloxanes catalyzed by a reusable palladium(II)/cationic bipyridyl system in water. Tetrahedron 2008, 64, 8164–8168. [Google Scholar] [CrossRef]
- Chen, S.N.; Wu, W.Y.; Tsai, F.Y. Homocoupling reaction of terminal alkynes catalyzed by a reusable cationic 2,2’-bipyridyl palladium(II)/CuI system in water. Green Chem. 2009, 11, 269–274. [Google Scholar] [CrossRef]
- Wang, Y.H.; Tsai, F.Y. Reusable rhodium(I)/cationic bipyridyl-catalyzed polymerization of phenylacetylenes in water under aerobic conditions. Chem. Lett. 2007, 1492–1493. [Google Scholar]
- Wu, W.Y.; Wang, J.C.; Tsai, F.Y. A reusable FeCl3.6H2O/cationic 2,2’-bipyridyl catalytic system for the coupling of aryl iodides with thiols in water under aerobic conditions. Green Chem. 2009, 11, 326–329. [Google Scholar] [CrossRef]
- Cui, X.; Li, Z.; Tao, C.Z.; Xu, Y.; Li, J.; Liu, L.; Guo, Q.X. N,N-Dimethyl-β-alanine as an inexpensive and efficient ligand for palladium-catalyzed Heck reaction. Org. Lett. 2006, 8, 2467–2470. [Google Scholar] [CrossRef]
- Mino, T.; Shirae, Y.; Sasai, Y.; Sakamoto, M.; Fujita, T. Phosphine-free palladium catalyzed Mizoroki−Heck reaction using hydrazone as a ligand. J. Org. Chem. 2006, 71, 6834–6839. [Google Scholar] [CrossRef]
- Aksın, Ö.; Türkmen, H.; Artok, L.; Çetinkaya, B.; Ni, C.; Büyükgüngör, O.; Özkal, E. Effect of immobilization on catalytic characteristics of saturated Pd-N-heterocyclic carbenes in Mizoroki–Heck reactions. J. Organomet. Chem. 2006, 691, 3027–3036. [Google Scholar]
- Chen, T.; Gao, J.; Shi, M. A novel tridentate NHC–Pd(II) complex and its application in the Suzuki and Heck-type cross-coupling reactions. Tetrahedron 2006, 62, 6289–6294. [Google Scholar] [CrossRef]
- Venkateswarlu, S.; Ramachandra, M.S.; Krishnaraju, A.V.; Trimurtulu, G.; Subbaraju, G.V. Antioxidant and antimicrobial activity evaluation of polyhydroxycinnamic acid ester derivatives. Indian J. Chem. Sect B 2006, 45, 252–257. [Google Scholar]
- Kondolff, I.; Feuerstein, M.; Doucet, H.; Santelli, M. Synthesis of all-cis-3-(2-diphenylphosphinoethyl)-1,2,4-tris(diphenylphosphinomethyl)cyclopentane (Ditricyp) from dicyclopentadiene. Tetrahedron 2007, 63, 9514–9521. [Google Scholar] [CrossRef]
- Andappan, M.M.S.; Nilsson, P.; Larhed, M. Arylboronic acids as versatile coupling partners in fast microwave promoted oxidative Heck chemistry. Mol. Divers. 2003, 7, 97–106. [Google Scholar] [CrossRef]
- Zeitler, K. Stereoselective synthesis of (E)-α,β-unsaturated esters via carbene-catalyzed redox esterification. Org. Lett. 2006, 8, 637–640. [Google Scholar] [CrossRef]
- Moreno-Mañas, M.; Pérez, M.; Pleixats, R. Stereospecific preparation of ethyl (E) and (Z)-3-aryl-3-phenylpropenoates by Heck reaction. Tetrahedron Lett. 1996, 37, 7449–7452. [Google Scholar] [CrossRef]
- Masllorens, J.; Moreno-Mañas, M.; Pla-Quintana, A.; Roglans, A. First Heck reaction with arenediazonium cations with recovery of Pd-triolefinic macrocyclic catalyst. Org. Lett. 2003, 5, 1559–1561. [Google Scholar] [CrossRef]
- Mu, B.; Li, T.; Xu, W.; Zeng, G.; Liu, P.; Wu, Y. Synthesis, characterization, and applications in Heck and Suzuki coupling reactions of amphiphilic cyclopalladated ferrocenylimine. Tetrahedron 2007, 63, 11475–11488. [Google Scholar]
- Chen, Y.; Huang, L.; Ranade, M.A.; Zhang, X.P. Iron(III) and ruthenium(II) porphyrin complex-catalyzed selective olefination of aldehydes with ethyl diazoacetate. J. Org. Chem. 2003, 68, 3714–3717. [Google Scholar] [CrossRef]
- Buchynskyy, A.; Stembera, K.; Hennig, L.; Findeisen, M.; Giesa, S.; Welzel, P. A Method for the introduction of reporter groups into moenomycin A, based on thiouronium salt chemistry. Eur. J. Org. Chem. 2002, 1163–1174. [Google Scholar]
- Zhou, P.; Li, Y.; Sun, P.; Zhou, J.; Bao, J. A novel Heck reaction catalyzed by Co hollow nanospheres in ligand-free condition. Chem. Commun. 2007, 1418–1420. [Google Scholar]
- Li, J.H.; Li, J.L.; Wang, D.P.; Pi, S.F.; Xie, Y.X.; Zhang, M.B.; Hu, X.C. CuI-catalyzed Suzuki−Miyaura and Sonogashira cross-coupling reactions using DABCO as ligand. J. Org. Chem. 2007, 72, 2053–2057. [Google Scholar]
- Iyer, S.; Kulkarni, G.M.; Ramesh, C. Mizoroki–Heck reaction, catalysis by nitrogen ligand Pd complexes and activation of aryl bromides. Tetrahedron 2004, 60, 2163–2172. [Google Scholar] [CrossRef]
- Hunter, D.H.; Cram, D.J. Electrophilic substitution at saturated carbon. XXVIII. The stereochemical capabilities of vinyl anions1. J. Am. Chem. Soc. 1966, 88, 5765–5776. [Google Scholar] [CrossRef]
- Nakao, Y.; Imanaka, H.; Chen, J.; Yada, A.; Hiyama, T. Synthesis and cross-coupling reaction of alkenyl[(2-hydroxymethyl)phenyl]dimethylsilanes. J. Organomet. Chem. 2007, 692, 585–603. [Google Scholar]
- Wheeler, O.H.; de Pabon, H.N.B. Synthesis of stilbenes. A comparative study1. J. Org. Chem. 1965, 30, 1473–1477. [Google Scholar] [CrossRef]
- Güsten, H.; Salzwedel, M. Die kernmagnetischen resonanzpektren substituierter cis-stilbene. Tetrahedron 1967, 23, 173–185. [Google Scholar] [CrossRef]
- Wang, L.; Li, H.; Li, P. Task-specific ionic liquid as base, ligand and reaction medium for the palladium-catalyzed Heck reaction. Tetrahedron 2009, 65, 364–368. [Google Scholar] [CrossRef]
- Wood, C.S.; Mallory, F.B. Photochemistry of stilbenes. IV. The preparation of substituted phenanthrenes1a–c. J. Org. Chem. 1964, 29, 3373–3377. [Google Scholar]
- Truce, W.E.; Lin, C.I.M. Stereoselective sulfene-tropone cycloadditions and stereospecific thermolysis of resulting adducts. J. Am. Chem. Soc. 1973, 95, 4426–4428. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Huang, S.-H.; Chen, J.-R.; Tsai, F.-Y. Palladium(II)/Cationic 2,2’-Bipyridyl System as a Highly Efficient and Reusable Catalyst for the Mizoroki-Heck Reaction in Water. Molecules 2010, 15, 315-330. https://doi.org/10.3390/molecules15010315
Huang S-H, Chen J-R, Tsai F-Y. Palladium(II)/Cationic 2,2’-Bipyridyl System as a Highly Efficient and Reusable Catalyst for the Mizoroki-Heck Reaction in Water. Molecules. 2010; 15(1):315-330. https://doi.org/10.3390/molecules15010315
Chicago/Turabian StyleHuang, Shao-Hsien, Jun-Rong Chen, and Fu-Yu Tsai. 2010. "Palladium(II)/Cationic 2,2’-Bipyridyl System as a Highly Efficient and Reusable Catalyst for the Mizoroki-Heck Reaction in Water" Molecules 15, no. 1: 315-330. https://doi.org/10.3390/molecules15010315





